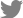|
 |
| jbm > Volume 30(4); 2023 > Article |
|
Abstract
Background
Spinal muscular atrophy (SMA) is a group of rare, inherited neuromuscular disorders. Bone health is often a neglected issue in children with SMA. This study aimed to evaluate the bone health status of children with SMA in Hong Kong.
Methods
This retrospective study included children with SMA who were managed in the Neuromuscular Disorder Clinics of 2 quaternary centers in Hong Kong. Bone health status was assessed by fracture history, bone mineral density (BMD) measured by dual energy X-ray absorptiometry, and serum 25-hydroxy-vitamin D (25[OH]D) level.
Results
Thirty-two children were included (males, 12). The median age was 10.8 years. BMD assessments were performed in 17 patients (SMA type 1=2, type 2=8, type 3=7). Low BMD was observed in 16 out of 17 patients. Four had a history of long bone fractures and were started on bisphosphonates. SMA types, age at last visit, sex, ambulation, and 25(OH)D level were not associated with fracture history or BMD Z-scores. Only one fulfilled the 2019 International Society for Clinical Densitometry (ISCD) pediatric definition of osteoporosis, with both low BMD and a history of clinically significant fracture.
Spinal muscular atrophy (SMA) is a group of rare inherited neuromuscular disorders with an incidence of around 10 in 100,000 live births.[1] It is caused by mutations in the survival of motor neuron 1 gene (SMN1) and is characterized by progressive muscle weakness and muscle atrophy. Childhood-onset SMA is classified into 3 subtypes based on the age of onset and best motor function achieved, with SMA type 1 being the most severe form (onset before the age of 6 months and non-sitter) and type 3 being the mildest form (walker but progressive muscle weakness after the age of 18 months).[1]
In the past, bone health was often a neglected issue in the care of children with SMA. However, osteoporosis is indeed a common problem, and fractures have been reported to be highly prevalent at up to 38% of these children.[2] This is attributed to both immobility and the functional role for SMN protein in bone remodelling.[2,3] In fact, bone health is one of the key factors determining the quality of life of patients with SMA. Therefore, in recent years, bone health assessments have been highlighted as a crucial part of SMA management.[4] Moreover, with the emergence of various effective disease-modifying treatments, including nusinersen and risdiplam, the natural course of the disease has been transformed.[5] Therefore, to maximise the effect of these treatments, it is important to prevent and manage its associated comorbidity, including osteoporotic fractures.
The objective of the study is to evaluate the bone health status in children with SMA in Hong Kong.
This is a retrospective study. Children with molecularly confirmed SMA, who were managed in 2 quaternary centers, Queen Mary Hospital and Hong Kong Children’s Hospital from 1st April 2021 to 31st March 2022, were recruited.
Their demographic data and clinical information, including SMA types, current ambulatory status, history of fractures, vitamin D status, bone mineral density (BMD), as well as drug treatment were retrieved from electronic medical records. Their bone health status and factors associated with compromised bone health including SMA types and ambulatory status were analysed.
The study was approved by the Institutional Review Board of the University of Hong Kong/Hospital Authority Hong Kong West Cluster and Hong Kong Children’s Hospital Clinical Research Ethics Committee.
The diagnosis of the children with SMA participating in this study was confirmed by molecular testing with homozygous deletion in SMN1 in an accredited genetic diagnostic laboratory. All patients had a classical SMA phenotype with verification by the attending pediatric neurologist (SHC). Ambulatory statuses were categorized as ‘walker’, ‘assisted walker’, ‘sitter’, ‘assisted sitter’ and ‘non-sitter’.[6]
Areal BMD (aBMD) was measured by dual energy X-ray absorptiometry (DXA) using the Hologic Horizon A system (Hologic Inc., Bedford, MA, USA) at the total body less head (TBLH), postero-anterior lumbar spine (LS) and/or lateral distal femur (LDF),[7,8] depending on whether patients had any metallic implants or fixed contractures that would affect the scanning results and positioning. Calculations of Z-scores and height-adjusted Z-score were based on published reference data.[9] Collapsed vertebrae, if any, were excluded from aBMD measurement. Data on patients’ last DXA measurement were collected and analysed.
A low BMD for age was defined as a BMD Z-score of ≤−2.0. The diagnosis of osteoporosis in children was defined based on the 2019 International Society of Clinical Densitometry (ISCD) Official Positions as the presence of both a clinically significant fracture history (i.e., 2 or more long bone fractures by age 10 years or 3 or more long bone fractures by the age 19 years) and BMD Z-score ≤−2.0, or the finding of one or more vertebral compression fractures, regardless of BMD findings.[10]
Descriptive statistics, including mean, median, standard deviation and interquartile range (IQR), were used according to the normality of the data. Proportions or percentages were used to describe qualitative data. Independent samples t-test and Mann-Whitney U test were used for comparisons between 2 groups (low BMD vs. normal BMD, fractured vs. no fracture group). All statistical analyses were performed using Stata version 15.1 (StataCorp., College Station, TX, USA). A P-value less than 0.05 was considered to indicate statistical significance (2-tailed).
Thirty-two children were included (males, 12). Table 1 summarized their clinical characteristics. Eight had SMA type 1, 14 had SMA type 2 and 10 had SMA type 3. The median age was 10.8 years (IQR, 7.0-15.6 years). Twenty-six patients had been put on nusinersen for a median duration of 3.0 years (IQR, 2.0-4.0 years), while 7 patients had been on risdiplam for a median duration of 0.8 years (IQR, 0.7-0.9 years).
Thirty patients were taking vitamin D supplements (mean dose, 1,467±820 IU daily) and their mean 25-hydroxy-vitamin D (25[OH]D) level was 83.6±38.2 nmol/L. Only 3 patients had vitamin D insufficiency (range, 39-49 nmol/L).
BMD assessments by DXA were performed in 17 patients (All were on either nusinersen or risdiplam; nusinersen=11, risdiplam=1, nusinersen initially and changed to risdiplam later=3; SMA type 1=2, type 2=8, type 3=7). DXA was not performed in the remaining patients mainly because of significant fixed contractures hindering the positioning of scanning or parental refusal. When compared between the groups with or without DXA performed, there was no difference in age, sex, SMA types and ambulatory status.
The median BMD Z-score at TBLH was −5.0 (IQR, −5.5 to −3.5), LS was −2.1 (IQR, −2.7 to −0.4) and that at LDF R1, R2 and R3 were −3.2 (IQR, −4.0 to −2.9), −3.8 (IQR, −4.5 to −2.8) and −2.8 (IQR, −4.3 to −2.4) respectively (Table 2). Since all patients had a height Z-score of >−1, no height-adjustment on BMD Z-score was performed. A low BMD for age with Z-scores ≤−2 was observed in 16 out of the 17 patients with DXA performed. The only patient with a normal BMD for age had SMA type 3 with independent ambulation and was started on nusinersen 1.3 years ago. Only one patient fulfilled the 2019 ISCD definition of osteoporosis (3 episodes of long bone fracture by the age of 7 years).
Four SMA type 2 patients had a history of long bone fractures with trivial injury, e.g., during a change of diaper or physiotherapy (‘assisted sitter’=1, ‘sitter’=2, ‘assisted walker’=1). These patients were all put on bisphosphonates (zoledronic acid) when long bone fractures were healed. Bone turnover markers, including C-terminal telopeptide (CTX-I), N-terminal propeptide of type I collagen (P1NP) and alkaline phosphatase, were checked before the first bisphosphonate infusion when long bone fractures had been healed (patient 1 did not have the baseline CTX and P1NP measured due to inadequate blood sample collected), and they all had low or low normal bone turnover markers. All the 4 patients developed a high fever and 2 developed mild, asymptomatic hypocalcemia after the first infusion of bisphosphonates, but there were no other adverse effects in subsequent infusion (Table 3). The first patient also developed sputum retention after the first infusion, which was managed with vigorous chest physiotherapy. This patient had been given 8 infusions of zoledronic acid for 4 years with interval improvement in BMD Z-scores (Fig. 1). At the time of review, no further fractures have been observed among these patients after bisphosphonates treatment.
SMA types, age, sex, current state of ambulation, duration of disease-modifying treatment, and 25(OH)D level were not associated with fracture history or BMD Z-scores. In addition, BMD Z-scores were not associated with fracture history.
We described the bone health status of a group of children with SMA in Hong Kong. Like previous reports, low BMD for age and fractures were common among this group of children. However, only one fulfilled the 2019 ISCD pediatric definition of osteoporosis with low BMD for age (Z-scores ≤−2) and a clinically significant fracture history. Undoubtedly, the 2019 ISCD definition has successfully prevented overdiagnosis of osteoporosis and unnecessary treatment in healthy children who do not have true skeletal fragility. However, in the clinical context of children with immobility or exposure to osteotoxic medications, this definition could result in underdiagnosis, and subsequently, undertreatment of secondary osteoporosis. With the fact that even a single episode of long bone fracture could lead to permanent loss of ambulation, waiting for the second, even the third episode of long bone fracture would superfluously delay the initiation of treatment in these high-risk children. This raised the question of whether a special definition is needed for this high-risk group of children.
In the past, possibly related to the limited survival in patients with SMA, the issue of bone health was infrequently discussed. With the advent of disease-modifying treatments, which have enabled improved motor function, survival, and quality of life, there has been a paradigm shift in the overall care of children with SMA, with a greater focus on the associated comorbidities, including bone health. Indeed, fractures do not only lead to acute pain, but also potentially permanent loss of ambulation. Cast immobilization following fractures, especially if prolonged, could aggravate muscle wasting and bone loss and further speed up the disease progression. In the latest International Consensus of SMA care and management, importance of a multidisciplinary approach in the various aspects including bone health, has been highlighted, and regular bone health monitoring with annual DXA study and checking of 25(OH)D levels was recommended.[4] However, practical implementation in the real-world clinical setting could be challenging. In our cohort, only 17 out of 32 patients had a DXA study performed. The main reasons for not having a DXA study performed include difficulties in positioning due to contractures and parental refusal, as some do not prefer another hospital visit and are worried about the risk of additional investigatory procedures. Clearly, a better service model is needed to facilitate the delivery of this recommended multidisciplinary care.
When compared to other neuromuscular conditions like Duchenne muscular dystrophy (DMD), the bone health status of children with SMA is much under-reported. Table 4 summarises the published cohorts on the topic. The prevalence of low BMD, vitamin D deficiency and fractures observed in various cohorts varies, which is likely contributed by the heterogenous patients’ population with different SMA types motor functions across different studies, as well as different standards of care in terms of bone health monitoring.[2,3,11-14] This makes direct comparisons difficult. SMA types, phenotype severity, increasing age, motor function, serum 25(OH)D and parathyroid hormone level have been reported to be important factors influencing BMD in children with SMA.[2,3,14] We were not able to find any factors associated with low BMD or fracture, likely related to the small sample size of our study.
Unsurprisingly, in cohorts with larger sample sizes, children with SMA type 1 have been reported to have significantly lower BMD Z-scores at all skeletal sites than children with SMA type 2 or 3.[3] On one hand, muscle and bone function as one unit,[15] and hence it is to be expected that patients with more severe neurological phenotype would have weaker bones. On the other hand, the deficiency of SMN protein also has pronounced effects on BMD. In fact, it has been well described that children with SMA tend to have more severe reduction in BMD than in children with other neuromuscular conditions of similar level of mobility.[16] In mouse model, the SMN protein has been demonstrated to have important functional roles in skeletal development [17] and bone resorption activity by interacting with cellular signaling molecules such as osteoclast stimulatory factor 1 (OSTF1), which is involved in the indirect activation of osteoclasts.[18] Mouse models of human SMA were observed to have poorly developed caudal vertebra and pelvic bone fractures.[17] Abundant activated osteoclasts were also observed on the histochemical staining of vertebrae from SMA mice, consistent with increased bone resorption.[19] With a more severe SMA phenotype, the amount of SMN protein is also lower.[17,19] All in all, this accounts for the high prevalence of low BMDs and fractures among children with SMA, especially SMA type 1. Therefore, it is logical to deduce that, with disease-modifying treatment, the bone phenotype might also be modified and improved.
Previous studies have also shown that a significant proportion of children with SMA have high CTX levels, indicating higher bone resorption.[2,14] This could be explained by the functional roles of the SMN protein in bone resorption through the interaction with OSTF1.[18] However, this phenomenon was not observed in our 4 children with long bone fractures. Indeed, they all had low CTX and P1NP levels, indicating a low bone turnover state. This might reflect different extents of contribution of the SMN protein and immobility during different stages of the disease course.
Nasomyont et al. [20] described their experience in intravenous bisphosphonate therapy in 8 children with SMA. Similar to our experience, an acute initial reaction was common after the first infusion but not the subsequent infusions. Minimal acute adverse effects were observed, and the infusions were effective in reducing incidences of further fractures. However, an atypical femur fracture was observed in one patient, and hence caution is still needed.[20]
Interestingly, we observed a much higher mean LS Z-score than TBLH and LDF. This phenomenon was also observed in children with DMD.[21,22] This would be suggestive of more cortical bone defects than trabecular defects, as well as the differential weight-bearing effects on BMD at different skeletal sites — with some weight bearing at the spine but much less weight bearing at the lower limbs. Alternatively, it might also be attributed to the presence of artifacts induced by scoliosis, which is common among our children with SMA and may falsely increase the measured aBMD (Fig. 2). This highlights the complexity in DXA interpretation and limitations of aBMD assessment by DXA in this group of children. This also infers the need of exploring other novel imaging modalities e.g., peripheral quantitative computed tomography, as part of the bone health assessment.
Apart from long bone fractures, asymptomatic, undiagnosed vertebral fractures are indeed not uncommon and have been reported among young children with SMA at a prevalence of approximately 10%.[14] This underscores the importance of an annual vertebral fracture screening as part of the bone health assessment, which was not the standard of care in our practice before, but we have implemented this as our routine monitoring recently. In fact, with the recent commencement of service of the Hong Kong Children’s Hospital, a one-stop, multidisciplinary clinic with joint assessment by a pediatric neurologist, pediatric orthopedic surgeon, and pediatric endocrinologist has been set up since 2021. DXA scan is performed by dedicated radiographers experienced in handling children with medical complexities and fragile bones. In contrary to most centers DXA service is provided in the Radiology Department, we particularly designed the DXA service to be delivered in the ambulatory care center, which is just one floor above our clinic, with support from onsite pediatricians and nurses. All these arrangements would help reduce the anxiety of parents about the risk and inconvenience of additional investigatory procedures and facilitate better communication between different disciplines for comprehensive care delivery.
There are several limitations in this study. First, as a retrospective study, not all patients had undergone the bone health assessment with DXA. Patients’ diet history and calcium intake, as well as details on the skeletal geometry, were also not available, which could also contribute to their bone health status and fracture risk. The sample size was small. However, with limited publications on bone health status in this rare condition, this is still a valuable addition to the existing literature. It also provides further information on health service needs in this group of children for better planning of health care resources.
Low BMD and long bone fractures were common among children with SMA in Hong Kong but osteoporosis based on the 2019 ISCD definition was uncommon. This latest ISCD pediatric definition of osteoporosis may result in underdiagnosis in this group of at-risk children and could potentially delay appropriate treatment for osteoporosis. Despite clear recommendations on regular bone health surveillance, practical implementation in the real-world could be challenging. Modifications in the care delivery system, e.g., a one-stop multidisciplinary service model, would facilitate the practical implementation. Longer-term, prospective studies with larger sample sizes are needed to investigate the effect of disease-modifying treatment and bisphosphonates on skeletal phenotypes in this group of children.
Acknowledgments
The authors would like to thank all patients and families in joining this study. We would also like to thank Ms. Lee BY Rachel for her professional English editing support.
DECLARATIONS
Fig. 1
Serial changes in bone mineral density Z-score at total body less head (TBLH) in patient 1 after bisphosphonates and risdiplam.

Fig. 2
Discrepant areal bone mineral density (BMD) Z-score at different body sites due to scoliosis. DXA, dual energy X-ray absorptiometry; BMC, bone mineral content; AM, age-matched; CV, coefficient of variation; Lt., left; Rt., right; T, thoracic; L, lumbar.

Table 1
Summary of clinical characteristics of the 32 children with SMA
| No. | SMA type | Sex | Age (yr) | TBLH BMD (Z-score) | LS BMD (Z-score) | LDF R1 BMD (Z-score) | LDF R2 BMD (Z-score) | LDF R3 BMD (Z-score) | 25(OH)D (nmol/L) | Ambulatory status |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 1 | F | 5.8 | - | - | - | - | - | 70 | Sitter |
| 2 | 1 | F | 3.4 | - | - | - | - | - | 53 | Assisted walker |
| 3 | 1 | F | 22.5 | - | −2.8 | - | - | - | 49 | Assisted walker |
| 4 | 1 | M | 7.0 | −5.4 | −3.4 | −3.2 | −3.8 | −2.7 | 64 | Non-sitter |
| 5 | 1 | F | 8.3 | - | - | - | - | - | 63 | Non-sitter |
| 6 | 1 | M | 11.0 | −6.3 | −2.1 | −3.2 | −4.0 | −4.3 | 105 | Non-sitter |
| 7 | 1 | F | 6.9 | - | - | - | - | - | 39 | Sitter |
| 8 | 1 | F | 16.2 | - | - | - | - | - | 77 | Non-sitter |
| 9a) | 2 | M | 6.4 | −4.5 | −4.1 | −5.00 | −4.9 | −4.5 | 190 | Sitter |
| 10 | 2 | F | 11.1 | - | - | - | - | - | 210 | Sitter |
| 11 | 2 | F | 13.3 | - | - | - | - | - | 48 | Assisted walker |
| 12a) | 2 | F | 6.3 | −6.3 | −4.7 | −4.0 | −4.5 | −4.5 | 94 | Sitter |
| 13 | 2 | M | 7.8 | −5.0 | −2.3 | - | - | - | 75 | Assisted walker |
| 14 | 2 | F | 17.6 | - | - | −3.3 | −3.7 | −3.8 | 63 | Sitter |
| 15 | 2 | M | 19.4 | - | - | - | - | - | 67 | Non-sitter |
| 16 | 2 | M | 6.2 | - | - | - | - | - | 83 | Sitter |
| 17 | 2 | F | 14.4 | - | - | - | - | - | 60 | Sitter |
| 18 | 2 | F | 12.9 | −5.0 | −2.0 | −4.4 | −4.9 | −4.3 | 126 | Sitter |
| 19 | 2 | F | 6.3 | - | - | - | - | - | 86 | Sitter |
| 20 | 2 | M | 9.3 | −5.5 | −1.6 | −2.3 | −2.8 | −1.9 | 125 | Assisted walker |
| 21a) | 2 | M | 10.9 | −5.5 | −2.5 | −3.5 | −3.8 | −2.9 | 55 | Assisted walker |
| 22a) | 2 | F | 7.4 | −5.5 | - | −5.5 | −4.8 | −2.7 | 93 | Non-sitter |
| 23 | 3 | M | 9.2 | −1.0 | 0.8 | −2.0 | −2.9 | −2.4 | 66 | Assisted walker |
| 24 | 3 | F | 19.2 | −3.7 | 1.0 | −3.0 | −2.8 | −0.9 | 56 | Sitter |
| 25 | 3 | F | 18.0 | - | - | - | - | - | 59 | Walker |
| 26 | 3 | M | 15.9 | −5.3 | −0.6 | −2.9 | −2.6 | −2.8 | 67 | Assisted walker |
| 27 | 3 | F | 18.3 | −1.5 | −0.4 | - | - | - | 63 | Walker |
| 28 | 3 | F | 15.3 | −3.3 | −0.3 | - | - | - | 85 | Assisted walker |
| 29 | 3 | M | 10.8 | - | - | - | - | - | 136 | Walker |
| 30 | 3 | F | 8.9 | −5.0 | −2.5 | - | - | - | 76 | Walker |
| 31 | 3 | M | 10.7 | −2.5 | 0.1 | 0.2 | 0.4 | 0.5 | 91 | Walker |
| 32 | 3 | F | 3.1 | - | - | - | - | - | 80 | Assisted walker |
Table 2
Summary of BMD Z-score at different skeletal sites
| DXA sites | Median BMD Z-score (IQR) |
|---|---|
| TBLH | −5.0 (−5.5 to −3.5) |
| LS | −2.0 (−2.5 to −0.3) |
| LDF R1 | −3.2 (−4.0 to −2.9) |
| LDF R2 | −3.8 (−4.5 to −2.8) |
| LDF R3 | −2.8 (−4.3 to −2.4) |
Table 3
Baseline clinical characteristics of the 4 patients with long bone fracture before treatment with zoledronic acid (after long bone fractures healed)
Table 4
Summary of different cohorts of bone health status of children with SMA
| References | Year | SMA types | Sample size | Mean age (yr) | Prevalence of fracture | Prevalence of low BMD | Prevalence of VDDa) | Factors associated with low BMDb) |
|---|---|---|---|---|---|---|---|---|
| Kinali et al. [12] | 2004 | 2, 3 | 12 | 11.8 | Not reported | 16.7% | Not reported | Older age |
| Aton et al. [13] | 2014 | 1 | 40 | 1.6 | Not reported | NA | 75.0% | Low vitamin D and calcium intakes |
| Vai et al. [2] | 2015 | 2, 3 | 30 | 1.3-14.3 | 36.7% | 50.0%c) | 6.7% | |
| Wasserman et al. [3] | 2017 | 1, 2, 3 | 85 | 1.0-18.0 | 38.0% | 85.0% | Not reported | Not reported |
| Baranello et al. [14] | 2019 | 2, 3 | 32 | 3.3 | 28.1% | 37.5% | 31.3% VDI | PTH, 25(OH)D levels |
| Peng et al. [11] | 2021 | 2, 3 | 40 | 5.4 | 5.0% | 67.5% | Not reported | SMA types, PTH levels |
| Current study | 2023 | 1, 2, 3 | 32 | 10.8 (median) | 12.5% | 93.3%d) |
0.0% VDD 9.4% VDI |
Non-statistically significant |
REFERENCES
1. Mercuri E, Bertini E, Iannaccone ST. Childhood spinal muscular atrophy: controversies and challenges. Lancet Neurol 2012;11:443-52.
https://doi.org/10.1016/s1474-4422(12)70061-3.


2. Vai S, Bianchi ML, Moroni I, et al. Bone and spinal muscular atrophy. Bone 2015;79:116-20.
https://doi.org/10.1016/j.bone.2015.05.039.


3. Wasserman HM, Hornung LN, Stenger PJ, et al. Low bone mineral density and fractures are highly prevalent in pediatric patients with spinal muscular atrophy regardless of disease severity. Neuromuscul Disord 2017;27:331-7.
https://doi.org/10.1016/j.nmd.2017.01.019.



4. Mercuri E, Finkel RS, Muntoni F, et al. Diagnosis and management of spinal muscular atrophy: part 1: recommendations for diagnosis, rehabilitation, orthopedic and nutritional care. Neuromuscul Disord 2018;28:103-15.
https://doi.org/10.1016/j.nmd.2017.11.005.


5. Schorling DC, Pechmann A, Kirschner J. Advances in treatment of spinal muscular atrophy - new phenotypes, new challenges, new implications for care. J Neuromuscul Dis 2020;7:1-13.
https://doi.org/10.3233/jnd-190424.



6. Pierzchlewicz K, Kępa I, Podogrodzki J, et al. Spinal muscular atrophy: the use of functional motor scales in the era of disease-modifying treatment. Child Neurol Open 2021;8:2329048x211008725..
https://doi.org/10.1177/2329048x211008725.



7. Mueske NM, Chan LS, Wren TA. Reliability of lateral distal femur dual-energy X-ray absorptiometry measures. J Clin Densitom 2014;17:522-7.
https://doi.org/10.1016/j.jocd.2013.02.010.



8. Weber DR, Boyce A, Gordon C, et al. The utility of DXA assessment at the forearm, proximal femur, and lateral distal femur, and vertebral fracture assessment in the pediatric population: 2019 ISCD official position. J Clin Densitom 2019;22:567-89.
https://doi.org/10.1016/j.jocd.2019.07.002.



9. Zemel BS, Kalkwarf HJ, Gilsanz V, et al. Revised reference curves for bone mineral content and areal bone mineral density according to age and sex for black and non-black children: results of the bone mineral density in childhood study. J Clin Endocrinol Metab 2011;96:3160-9.
https://doi.org/10.1210/jc.2011-1111.



10. Shuhart CR, Yeap SS, Anderson PA, et al. Executive summary of the 2019 ISCD position development conference on monitoring treatment, DXA cross-calibration and least significant change, spinal cord injury, peri-prosthetic and orthopedic bone health, transgender medicine, and pediatrics. J Clin Densitom 2019;22:453-71.
https://doi.org/10.1016/j.jocd.2019.07.001.


11. Peng X, Qu Y, Li X, et al. Bone mineral density and its influencing factors in Chinese children with spinal muscular atrophy types 2 and 3. BMC Musculoskelet Disord 2021;22:729.
https://doi.org/10.1186/s12891-021-04613-x.



12. Kinali M, Banks LM, Mercuri E, et al. Bone mineral density in a paediatric spinal muscular atrophy population. Neuropediatrics 2004;35:325-8.
https://doi.org/10.1055/s-2004-830366.


13. Aton J, Davis RH, Jordan KC, et al. Vitamin D intake is inadequate in spinal muscular atrophy type I cohort: correlations with bone health. J Child Neurol 2014;29:374-80.
https://doi.org/10.1177/0883073812471857.



14. Baranello G, Vai S, Broggi F, et al. Evolution of bone mineral density, bone metabolism and fragility fractures in Spinal Muscular Atrophy (SMA) types 2 and 3. Neuromuscul Disord 2019;29:525-32.
https://doi.org/10.1016/j.nmd.2019.06.001.


15. Veilleux LN, Rauch F. Muscle-bone interactions in pediatric bone diseases. Curr Osteoporos Rep 2017;15:425-32.
https://doi.org/10.1007/s11914-017-0396-6.


16. Khatri IA, Chaudhry US, Seikaly MG, et al. Low bone mineral density in spinal muscular atrophy. J Clin Neuromuscul Dis 2008;10:11-7.
https://doi.org/10.1097/CND.0b013e318183e0fa.


17. Shanmugarajan S, Swoboda KJ, Iannaccone ST, et al. Congenital bone fractures in spinal muscular atrophy: functional role for SMN protein in bone remodeling. J Child Neurol 2007;22:967-73.
https://doi.org/10.1177/0883073807305664.



18. Vermeren M, Lyraki R, Wani S, et al. Osteoclast stimulation factor 1 (Ostf1) KNOCKOUT increases trabecular bone mass in mice. Mamm Genome 2017;28:498-514.
https://doi.org/10.1007/s00335-017-9718-3.



19. Shanmugarajan S, Tsuruga E, Swoboda KJ, et al. Bone loss in survival motor neuron (Smn(−/−) SMN2) genetic mouse model of spinal muscular atrophy. J Pathol 2009;219:52-60.
https://doi.org/10.1002/path.2566.



20. Nasomyont N, Hornung LN, Wasserman H. Intravenous bisphosphonate therapy in children with spinal muscular atrophy. Osteoporos Int 2020;31:995-1000.
https://doi.org/10.1007/s00198-019-05227-9.


21. Tung JY, Lam TP, Chan SH. Bone microarchitectural alterations in boys with Duchenne muscular dystrophy on long-term glucocorticoid treatment. J Bone Miner Metab 2021;39:606-11.
https://doi.org/10.1007/s00774-020-01196-w.


22. Tian C, Wong BL, Hornung L, et al. Bone health measures in glucocorticoid-treated ambulatory boys with Duchenne muscular dystrophy. Neuromuscul Disord 2016;26:760-7.
https://doi.org/10.1016/j.nmd.2016.08.011.


- TOOLS
-
METRICS

-
- 0 Crossref
- 0 Scopus
- 2,312 View
- 46 Download
- ORCID iDs
-
Joanna Yuet-Ling Tung

https://orcid.org/0000-0001-7897-716XTsz-Kit Chow

https://orcid.org/0000-0002-1736-8771Monique Wai

https://orcid.org/0000-0003-4462-6016Jasmine Lo

https://orcid.org/0000-0002-2191-8588Sophelia Hoi Shan Chan

https://orcid.org/0000-0002-2990-0163 - Related articles




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print