The Role of Titanium Particles and Ions in the Pathogenesis of Peri-Implantitis
Article information
Abstract
Titanium (Ti) particles and ions have been investigated in recent years as important factors in the pathogenesis of peri-implantitis. However, their role in the pathogenesis is yet to be fully understood. A review of pertinent literature was performed in various databases to determine the current position of Ti particles and ions role in the pathogenesis of peri-implantitis. There are several in vitro, preclinical and clinical published studies that have addressed the role of Ti particles and ions in the pathogenesis of peri-implantitis. These studies explored the effect of Ti particles and ions in the pathogenesis of peri-implantitis with respect to foreign body reaction, cellular response, epigenetic mechanisms, namely DNA methylation, and the oral microbiome. Studies have shown that the release of Ti particles/ions during implant insertion, early healing stages, late healing stages, and treatments during peri-implantitis might contribute to peri-implantitis through different mechanisms, such as foreign body reaction, cellular response, DNA methylation, and shaping the oral microbiome by increasing dysbiosis. However, further studies are needed to elucidate the complex interactions between all these mechanisms and Ti particles/ions in the pathogenesis and progression of peri-implantitis.
INTRODUCTION
During the last 55 years, osseointegration of dental implants has shown a success rate of above 90%, however, most complications related to dental implants occur after they have been fully osseointegrated and in function for a long period of time.[1–3] Peri-implant inflammation and infection were revealed to be significant causes for the late loss of osseointegrated implants.[4,5] The term “peri-implantitis” was described as an inflammatory process that causes marginal bone loss beyond the physiologic bone remodeling.[6] This inflammatory process is multifactorial and several components have been reported as promoters of peri-implantitis pathogenesis such as bacterial infection, mechanical stress, biocorrosion and fragments released from the dental implant surfaces.[7–9] These fragments released from the implant surface are classified as particles (particulate metal in a stable atomic electric condition) or ions (particulate metal in an unstable atomic electric condition), which impact the reaction of cells responsible for the inflammatory process and bone remodeling.[8,10–12]
Specifically, the release of Titanium (Ti) particles/ions derived from implant surfaces into the adjacent tissues has been demonstrated during the early and late stages of successful osseointegration through different processes.[10,11, 13] For example, modern surfaces have been developed to achieve faster osseointegration, however, implant insertion into the alveolar bone using high insertion torques might result in the rupture of metal bonds and the release of particles into the surrounding tissues.[14,15] Moreover, factors such as metal biocorrosion and mechanical stress caused by mechanical loading over implant-supported crowns lead to the release of ions and particles into the peri-implant tissues.[12,16] Lastly, therapeutic modalities applied to treat peri-implant diseases can result in a significant release of metal particles based on the technique applied to clean the implant surface.[17,18]
The influence of Ti particles on the surrounding tissues seems to be dependent on the size and surface properties of the particle.[19] In fact, Ti particles smaller than 10 μm have been reported as cytotoxic, causing different responses such as inflammation, foreign body reaction, epigenetics changes, and bone resorption.[19–22]
Several methods have been proposed for the decontamination of implant surfaces after diagnosis of peri-implantitis, anticipated to treat the disease.[23–25] However, different studies have demonstrated that decontamination techniques using burs or metal instruments, i.e., implantoplasty, generate Ti particles around the implant, which could have negative effects on bone regeneration.[26,27] As such, it must be highlighted that the concentration and size of these released particles during implantoplasty treatments are dependent on the implant chemical composition and implant surface modifications.[27]
To date, the role of the released Ti particles/ions in the pathogenesis and progression of peri-implantitis is still not fully understood. The aim of this concise narrative review is to highlight the effect of Ti particles/ions released into peri-implant tissues on the cellular and tissue responses, which might result in the pathogenesis and/or progression of peri-implantitis.
EFFECT OF TITANIUM PARTICLES & IONS IN INDUCING FOREIGN BODY REACTION RESPONSE IN PERI-IMPLANTITIS
In recent years, increased attention has been put on the understanding of the role of Ti particles and ions in eliciting a foreign body reaction. It has been suggested that peri-implant marginal bone loss due to peri-implantitis, might be attributed to a change in the foreign body equilibrium between the host immune system and the dental implant.[28]
1. In vitro & preclinical studies
In a rat study,[20] soft tissues around the coronal tip of implants, placed bilaterally in the maxillary first molar area, were injected with 20 mg Ti particles, to induce an aseptic foreign body reaction. In the test group, macrophages were further depleted by the local injection of liposomal clodronate (Ti + LipClod). Ti-injected rats treated with phosphate buffered saline (Ti + PBS) or empty liposome (Ti + Lip) served as controls. Eight weeks later, the animals were sacrificed and the harvested maxillae were subjected to gene expression analysis of macrophage markers and histological analysis of macrophage number, distribution, and phenotype. Bone level changes were analyzed radiologically.[20]
In comparison to both control groups, the Ti + LipClod group showed a significantly decreased macrophage density and bone loss around the implant. In fact, more macrophages infiltrated into peri-implant tissue in both control groups, predominantly M1 phenotype. On the contrary, macrophage density was reduced in the Ti + LipClod group, with M1 macrophages slightly more than M2. Accordingly, messenger RNA expression of tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), IL-6, and receptor activator of nuclear factor-κB ligand (RANKL) increased significantly in both control groups compared to the Ti + LipClod group.[20] Taking all these findings together, the authors suggested that Ti particles have a negative effect on peri-implant tissue by activating macrophages, mainly M1 phenotype, which promotes local secretion of inflammatory cytokines. As such, macrophage polarization can impact peri-implant bone loss caused by Ti particles, suggesting a foreign body reaction by these particles, eliciting a host immune response characterized by macrophage activation.[20] Nonetheless, the findings of this study need to be interpreted with caution, as overdosed Ti particles were used to induce acute aseptic inflammation, which does not reflect actual clinical circumstances in which trace wear particles are continuously released from the implant surface. Moreover, foreign body reaction is a complex process involving the interaction of various immune cells, and not only macrophages.[20]
In another rat study, Toledano-Serrabona and colleagues [29] evaluated mandibular bone defects in Sprague-Dawley rats, filled with metallic debris obtained by implantoplasty from Ti-6Al-4V dental implants.
Thirty days post-surgery, animals were euthanized and histological analysis revealed that metal particles were surrounded by a foreign body reaction characterized by the presence of histiocytes and multinucleated giant cells, suggesting that Ti particles triggered chronic inflammation in bone. Interestingly, there was also a high concentration of Ti particles in the spleen, liver and brain, indicating a systemic release and distant accumulation of Ti metal particles.
2. Human studies
A recently published study aimed at identifying Ti particles and histopathological characteristics in 39 peri-implantitis cases compared to 35 periodontitis cases from harvested biopsies containing granulation tissue.[30] Ti particles were identified using scanning electron microscopy (SEM) coupled with dispersive X-ray spectrometry. Histological analysis showed that peri-implantitis granulation tissue was characterized by intense neovascularization and the presence of a chronic inflammatory infiltrate dominated by plasma cells, neutrophils, and macrophages. In comparison to periodontitis, peri-implantitis samples displayed higher macrophage afflux and more expressed neovascularization based on immunohistochemistry of histological samples, suggesting a more severe inflammatory pattern in peri-implantitis. Furthermore, Ti particles were identified in all peri-implantitis samples, as free metal bodies interspersed within granulation tissue. Interestingly, macrophages or multinucleated giant cells engulfing Ti particles were absent in all the samples, suggesting an absence of foreign body reaction and a direct pathological effect of Ti particles.
In another study, 10 peri-implant soft tissue biopsies of patients diagnosed with peri-implantitis were obtained to identify the presence of Ti particles using SEM and dispersive X-ray spectrometry, and also to characterize inflammatory infiltrates suggestive of foreign body reaction.[31] Histological analysis, coupled with SEM and dispersive X-ray spectrometry identified Ti wear particles in 90% of samples in association with a chronic inflammatory infiltrate. Furthermore, significant overexpression of RANKL was observed with a trend of overexpression of transforming growth factor-β1 and IL-33 in areas of Ti.
These 2 studies [30,31] represent pioneering interrogations of the nature of peri-implantitis in humans. However, the observations still need to be interpreted with caution due to the cross-sectional nature of the studies, lack of information regarding the duration of peri-implantitis in recruited patients and the general qualitative assessment of infiltrating cells. Nevertheless, there is a great opportunity to gather valuable information in human specimens on the evolution of peri-implantitis in relation to implant material constituents.
EFFECT OF TITANIUM PARTICLES & IONS ON THE CELLULAR RESPONSE IN PERI-IMPLANTITIS
Several factors can stimulate the release of Ti particles/ions before or after peri-implantitis. However, most of the studies in literature have reported the presence of Ti particles after dental implants have been fully osseointegrated and in function for a long period of time.[8,12,13]
In the case of peri-implantitis, there is increased mechanical stress around the dental implant due to the treatment procedures performed at the clinic to treat the condition, and also due to the cleaning procedures performed by the patient to remove the bacterial biofilm. Mechanical stress stimulated by occlusion forces, mechanical treatments for peri-implantitis, and the presence of infection generating lower pHs around the dental surfaces were demonstrated as contributors to Ti particles/ions release,[8,19,22,26] suggesting the increased possibility of Ti particles/ions release after diagnosis of peri-implantitis.
As such, the released particles/ions can affect the cellular response in peri-implantitis. In this section, the effect of Ti particles/ions on the cellular responses, before (i.e., during implant placement) or after peri-implantitis has occurred, is reported.
1. In vitro & preclinical studies
A recent ex vivo study with porcine mandibles investigated the release of Ti particles during implant placement and the presence of particles inside the cells in the surrounding tissues.[32] The authors demonstrated significant differences in the number of particles released based on the design, surfaces and chemical composition of the implants. However, all the tested implant systems (Straumann® and BioHorizons®) were shown to release Ti particles during implant insertion. Further, internalization of Ti debris was evident in human gingival fibroblasts and RAW 264.7 macrophages for all the tested systems in vitro.[32]
In another in vitro study, Ti-particles isolated from implant surfaces were applied to oral epithelial cells in culture to investigate molecular reactions of these cells. The findings revealed that DNA damage response was triggered by the interaction with Ti particles, suggesting a possible disruption of epithelial hemostasis and potential impairment of the epithelial barrier.[33] Another study, using neutrophil cells and rats for in vivo experiments, reported that Ti particles smaller than 10 μm induce inflammation and cytotoxicity in neutrophils by the stimulation of TNF-α secretion and phagocytosis. The authors concluded that particles below 10 μm in size induce cytotoxicity mainly from engulfment by inflammatory cells.[34]
Furthermore, Wachi et al. [35], demonstrated in an in vitro study that Ti ions (concentrations above 11 ppm) might alter the activity of bone cells and gingival epithelium cells, by generating cytotoxic-like effects to the surrounding tissues, and thus stimulating deteriorating effects on peri-implant tissues that may be associated with the progression of peri-implantitis and alveolar bone resorption. In another study [36], human macrophages cultured with Ti particles in pre-determined concentrations were revealed to stimulate the secretion of IL-1β and promote secondary stimuli for proinflammatory reactions. The authors demonstrated that after the filtration of the Ti ions released in the culture medium applied and re-investigation, the pro-inflammatory signals disappeared, indicating the impact of Ti ions on the inflammatory reaction.[36]
Therefore, taking the previously presented results together, it is possible to identify various cell types around the osseointegrated implants, that are affected by both Ti particles and Ti ions directly or indirectly, generating the activation of the immune system and indicating a role of Ti particles/ions on the initiation/progression of mucositis and peri-implantitis (Fig. 1).
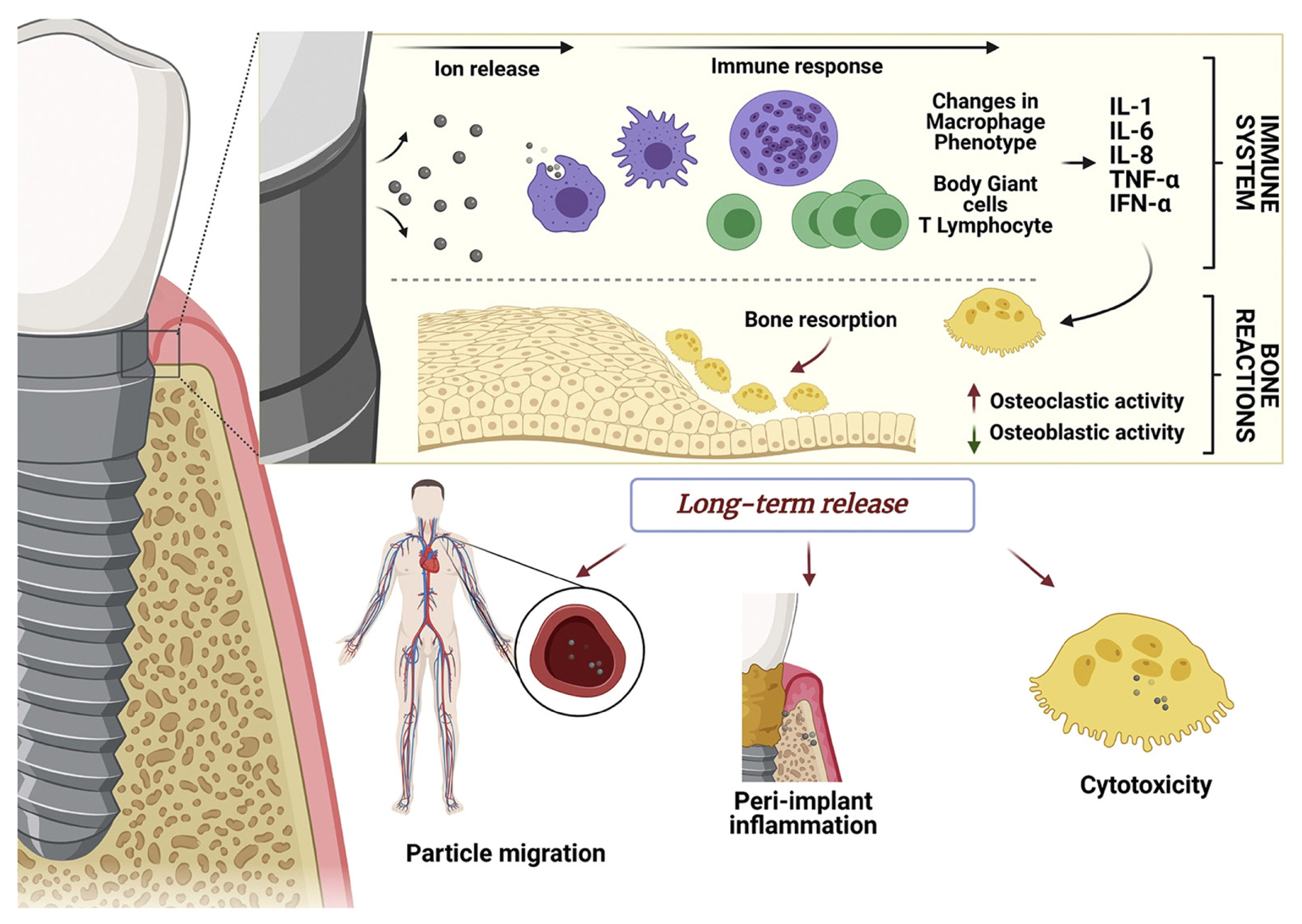
Schematic illustration showing tentative consequences promoted by ions/particles released from dental implants into the surrounding tissues. (1) At the early stages, the activation of immune response and bone resorption may be stimulated; (2) At late stages, the migration or accumulation of ions/particles might occur, resulting in peri-implant diseases. [Reprinted from “Innovative surfaces and alloys for dental implants: What about biointerface-safety concerns?”, by Kunrath MF, et al., 2021, Dent Mater, 37, pp. 1447–1462. Copyright 2021 by the Elsevier. Reprinted with permission].
Different techniques applied to decontaminate the implant surface, also known as implantoplasty, due to peri-implantitis infection have shown promising results, resulting in bone regeneration in the previously destructed area.[17,23,24] Nonetheless, some instruments employed in this decontamination procedure appear to promote a large-scale release of metal particles into the surrounding tissues.[17,26,27] For example, Eger et al. [37] demonstrated that the treatment of peri-implantitis using ultrasonic metal tips promoted the release of Ti particles from different implant surfaces. Moreover, the authors reported that these particles significantly increased the inflammatory and osteoclastogenic responses in vitro and in vivo, suggesting the possibility of aggravation of peri-implantitis, in short-term follow-ups (24–48 hr) and indicating the necessity of long-term examination in further studies to investigate the chronic effects of this exposure.[37] Current surfaces developed for dental implants with micro- or nano-textures, aiming for faster osseointegration, possess a sensitive morphology compared to smooth surfaces [38,39] and the utilization of some instruments for surface cleaning might facilitate the release of particles or ions. On the other hand, surface treatments applying subtractive methods such as etching, blasting, and anodizing, promote surface properties with higher corrosion resistance compared to additive-coated surfaces, suggesting lower rates of particle release during the biocorrosion process.[40,41] Results by Schwarz et al. [42] revealed that implantoplasty on modified surfaces by using diamond burs promoted the release of Ti particles from all tested samples, which might be linked to inflammation. Thus, techniques using less aggressive approaches, e.g., with non-metallic instruments, to treat dental implant surfaces affected by peri-implantitis might reduce the possibility of Ti particles release.
2. Human studies
Recently, some studies have investigated the effects of Ti particles/ions on the alveolar bone and surrounding tissues from human peri-implant tissue samples. Olmedo et al. [43] collected cytologic samples of the peri-implant mucosa, using a microbrush, from 30 patients with or without peri-implantitis to assess the presence of metal-like particles and Ti particles/ions in the surrounding tissues. Their results revealed that metal-like/Ti particles were released from the surface into the biologic milieu, regardless of the peri-implant status; however, the concentration of particles found was higher in the peri-implantitis group compared to the group without disease. Furthermore, the authors discussed the significance of the nature, size and concentration of these particles, correlating the level of reaction to these factors and to the time of biotribocorrosion process around implants made of Ti alloy. These results reported by the authors regarding the appearance of metal-like particles support the influence of metal particles in non-disease and disease conditions due to the corrosion of dental surfaces.[43] In a prospective human study, bone and mucosal samples were collected from patients with implants indicated for explantation due to peri-implantitis.[44] After analysis of the human samples removed close to the implant surfaces, Fretwurst et al. [44] found that 75% of the samples collected were presenting Ti or iron elements in their chemical composition, concluding that further immunohistochemical studies should be performed in order to clarify the relation between Ti particles and the progress of peri-implantitis.
Additionally, a study that utilized soft-tissue biopsies and peri-implant fluid samples collected close to implants presenting healthy, peri-implant mucositis and peri-implantitis conditions investigated the concentration of Ti particles using inductively coupled plasma mass spectrometry.[36] Their findings showed a variation in the Ti particle concentration between 7.3 and 38.9 μM in all the samples collected from the patients. Furthermore, Ti particle concentrations found in peri-implant mucosa revealed similar levels of metal (Ti particles) to concentrations (25–50 μM) that induced the activation and secretion of IL-1β in vitro, which might suggest the possibility of pro-inflammatory responses derived due to these elevated concentrations in some samples.[36]
Wilson et al. [45] reported the presence of Ti particles in human soft tissue biopsies collected from gingival flaps close to implants with peri-implantitis. The authors suggested that the presence of these metallic elements was promoted by implant corrosion during the implant function in the mouth. Moreover, the authors applied histology and energy-dispersive X-ray spectrometry to analyze the samples and demonstrated that these particles in the soft tissue were surrounded by chronic inflammation, suggesting that further studies should be designed to clarify the role of Ti fragments in peri-implantitis.[45]
Despite the fact that the previously presented human studies indicate that Ti particles/ions might play a role in activating inflammatory responses in mucositis and peri-implantitis, they do not totally elucidate the effect of Ti particles/ions in stimulating the progression of peri-implantitis. Another unclear phenomenon is the potential of particle migration in the human body; some reports have shown the possibility of particle release and migration in the blood and surrounding tissues to other distant tissues, suggesting a potential for metal bioaccumulation and cytotoxicity.[10,15,46] The in vitro and preclinical studies have demonstrated direct and indirect effects of Ti particles/ions in specific cell reactions investigated in short periods of time; meanwhile, human studies have shown the presence of Ti particles/ions in tissue samples collected close to implants with mucositis, peri-implantitis and healthy conditions in significant concentrations, however, no long-term studies are available to state the chronic effects of these metal fragments on the peri-implantitis progression.
EFFECT OF TITANIUM PARTICLES ON THE EPIGENETICS IN PERI-IMPLANTITIS
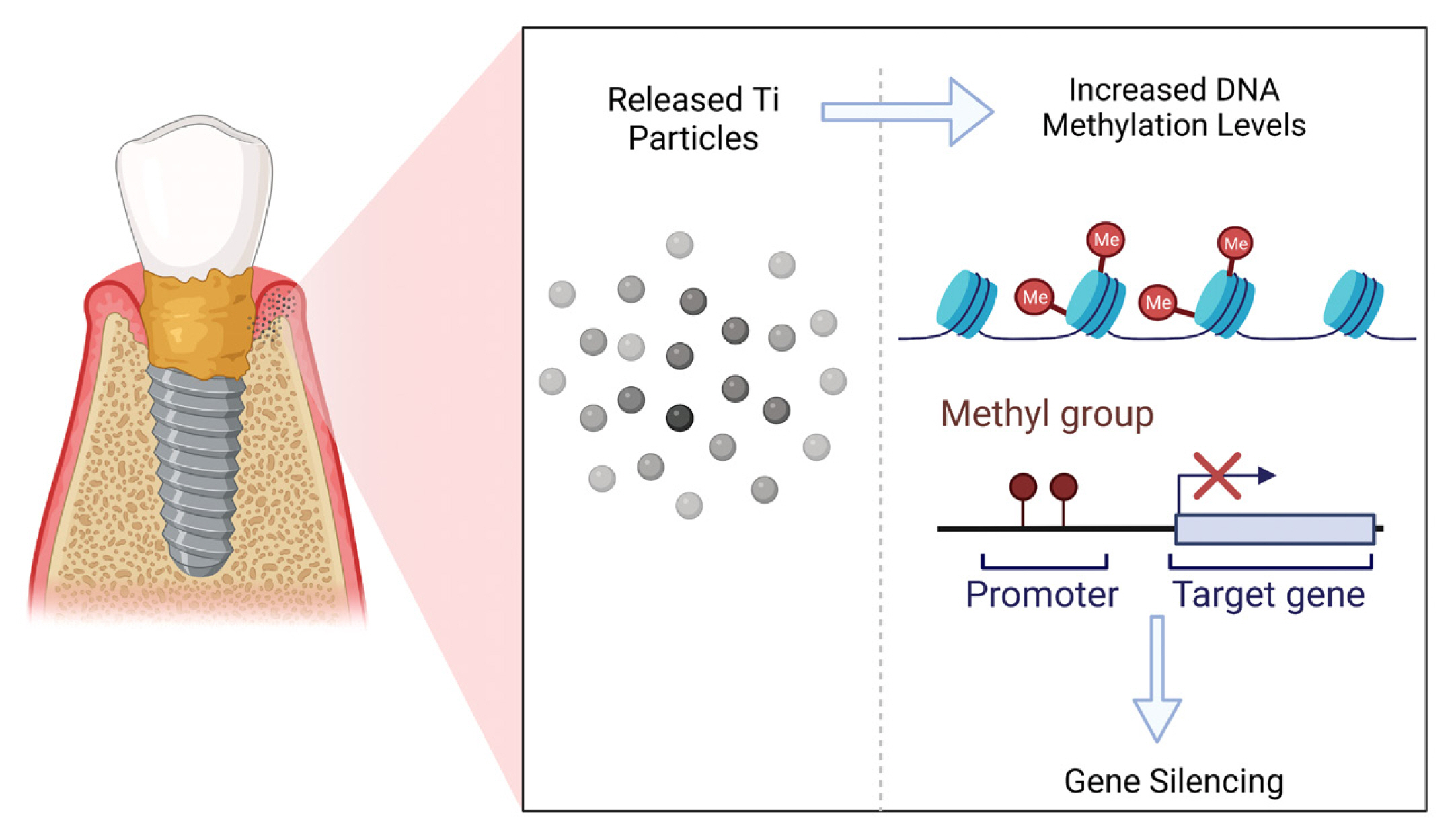
Epigenetic changes refer to alterations in the gene expression that are not encoded in the DNA sequence.[47,48] These epigenetic changes influence the chromatin configuration through chemical alteration of the DNA and its associated proteins called histones. To date, DNA methylation is the most studied epigenetic mechanism, in which methyl groups are added to the DNA, by specific enzymes, namely DNA methyl transferases, which results in gene silencing i.e., inactivation of gene expression. On the other hand, when these methyl groups are removed, in a process termed DNA demethylation, the gene expression is activated.[21]
Epigenetic changes are influenced by various environmental factors, such as diet, smoking, and bacterial biofilm.[49] As such, epigenetic changes are reversible when these factors are adjusted.
1. Human studies
To date, there are only 2 published human studies that have investigated the relationship between epigenetics, namely DNA methylation and peri-implantitis.[50,51] In one of these studies, the effect of Ti particles on the global DNA methylation levels was assessed in the peri-implant crevicular fluid (PICF) collected from peri-implantitis and controls.[50] Interestingly, the results of this study revealed that PICF with higher Ti quantities displayed significantly higher global DNA methylation levels, suggesting that the dissolution of Ti particles affects DNA methylation. However, more in-depth investigation is still needed before concluding a causal association between Ti particles and DNA methylation, as it is important to elucidate which genes are inactivated by this elevated DNA methylation, for example, genes associated with pro-inflammatory/anti-inflammatory cytokines, and genes associated with osteogenesis/osteoclastogenesis. Furthermore, the effect of Ti particles and ions, released by either minimally-rough or smooth Ti surfaces, on peri-implant tissues needs to be evaluated, since a recent in vitro study demonstrated that smooth and moderately rough Ti surfaces affect epigenetic changes differently,[52] which were correlated with lower DNA damage/repair markers.
In summary, more detailed studies are needed to investigate the relationship between Ti particles and DNA methylation, taking into account the type of implant surface and evaluating the DNA methylation changes of a specific gene panel not only in PICF but also in peri-implant mucosa and bone.
The effect of Ti particle release on DNA methylation in peri-implantitis is schematically illustrated in Figure 2.
EFFECT OF TITANIUM PARTICLES ON THE MICROBIOME IN PERI-IMPLANTITIS
To date, there are 2 published studies that have investigated the effect of Ti particles on the oral microbiome in peri-implantitis, because as known, peri-implantitis is mainly induced by the bacterial biofilm.[53]
1. In vitro & preclinical studies
In a recent study by Souza and colleagues [54], volunteers wore a palatal appliance, which contained Ti discs for 7 days, allowing for bacterial biofilm formation. After the collection of discs, the biofilms were treated in vitro as follows: (1) Ti particles (0.75% and 1%); (2) Ti ions (10 and 20 ppm); (3) a combination of 1% Ti particles and 20 ppm Ti ions. After 24 hr, biofilms were analyzed by checkboard DNA-DNA hybridization. Results showed that Ti particles affected the biofilm composition by increasing the population of 4 bacterial species (Streptococcus anginosus, Prevotella nigrescens, Capnocytophaga sputigena, and Actinomyces israelli) (P<0.05), while Ti ions resulted in higher levels of pathogens from the orange complex, which is disease-associated complex, and a reduction of species from the yellow complex, which is health-associated complex (P<0.05), suggesting that Ti ions released from dental implants might favor the growth of peri-implant pathogenic species. Interestingly, the combination of Ti particles and ions resulted in increased green complex (health-associated) and reduced yellow complex proportions (P<0.05). The lack of a synergistic effect of this combination on bacterial complexes might be attributed to the reduction of Ti ions precipitation in the biofilm due to the saturation of the extracellular environment with Ti particles and also due to the fact that an increase of anaerobic bacteria implicated in pathogenesis was only found for the Ti ions group and not the Ti particles group.
Therefore, the presence of Ti dissolution products, especially Ti ions, around dental implants might result in microbial dysbiosis and eventually, peri-implantitis.
2. Human studies
In a human study, Daubert et al. [55] assessed the effect of Ti particles on the oral microbiome in subgingival bacterial biofilm collected from 9 patients with fifteen implants of 10 years in function. A subgingival plaque was collected from 9 healthy implants and 6 implants affected by peri-implantitis. The oral microbiome was profiled using 16S ribosomal RNA next-generation sequencing, and Ti particles were quantified by mass spectrometry. Results of this study showed that Ti was associated with: the peri-implant disease status (P<0.02), the microbiota composition (P<0.045), and correlated with the first principal component of the microbiome (rho=0.552) and its alpha-diversity (rho=−0.496). As such, it seems that Ti dissolution products modify the peri-implant microbiota via enrichment for selected group of bacteria.
CONCLUSIONS
Several studies have shown that the released Ti particles/ions might contribute to peri-implantitis through different mechanisms, such as foreign body reaction, cellular response of epithelial, gingival, inflammatory, and bone cells, epigenetic mechanisms namely DNA methylation and shaping the oral microbiome by favoring dysbiosis. The concentration and size of these particles/ions depend on the implant’s chemical composition and surface treatment applied to the dental implant. However, further studies are urgently required to elucidate the complex interactions between all these mechanisms and Ti particles/ions in the pathogenesis and progression of peri-implantitis.
Notes
Funding
The study was supported by grants from the Swedish Research Council (2018-02891), the Swedish state under the agreement between the Swedish government and the county councils, the ALF agreement (ALFGBG-725641), the IngaBritt and Arne Lundberg Foundation, the Sylvan Foundation and the Area of Advance Materials of Chalmers and GU Biomaterials within the Strategic Research Area initiative launched by the Swedish government. MFK is supported by the Osteology research scholarship (Osteology Foundation, Lucerne, Switzerland). For this study, MFK also received additional funding from the Hjalmar Svensson Foundation (Grant number: HJSV2022015).
Ethics approval and consent to participate
Not applicable.
Conflict of interest
No potential conflict of interest relevant to this article was reported. Figure 2 was created with BioRender software (https://biorender.com) (accessed on 16 Jul 2022).