A Case of Low Bone Mineral Density with Vitamin D Deficiency Due to Prolonged Lactation and Severe Malnutrition
Article information
Abstract
Malnutrition associated vitamin D deficiency contributes to the calcium loss from bone and results in osteoporosis and osteomalacia at final stage. Osteomalacia is characterized with softening of bone secondary to defective bone mineralization. Here, we report a case of possible osteomalacia caused by prolonged lactation and severe malnutrition in 35-year-old female. She was a housewife and her body mass index was 11.8 kg/m2. She was diagnosed with severe osteoporosis in regular health check-up 2 years ago, but did not take any medication. Nine months ago, she had been treated with anti-tuberculosis medications for 6 month due to active pulmonary tuberculosis. After complete remission of pulmonary tuberculosis, she had lost her appetite severely. Furthermore, she felt gait difficulty and suffered from generalized bone pain. On serologic examination, hypocalcemia, hypophosphatemia, high alkaline phosphatase, low vitamin D3 and high parathyroid hormone level were seen. In the bone mineral density, Z-score from her lumbar spine was -6.5. She was treated with oral calcium and vitamin D3 intramuscularly. After 1 year treatment, she felt significant improvement in bone pain and could walk alone. Also her serum calcium, phosphate and vitamin D3 level are all normalized.
INTRODUCTION
Osteomalacia is an end stage bone disease characterized by impaired mineralization of bone matrix resulted in softening of bone and skeletal deformities.[1] There are 4 types of osteomalacia; osteomalacia with normal calcium and phosphorus metabolism; osteomalacia with altered metabolism of vitamin D; hypophosphatemic osteomalacia; and osteomalacia with vitamin D deficiency.[2] Among these, osteomalacia with vitamin D deficiency is the most common type and is characterized with low levels of serum vitamin D, calcium, phosphorus and elevated parathyroid hormone (PTH).
Vitamin D has 2 forms and several metabolites. Respectively, there are 2 forms, vitamin D2 and vitamin D3, also called ergocalciferol and cholecalciferol. Vitamin D3 is produced in the skin by the ultraviolet B radiation from sunlight or is absorbed by the diet. Also vitamin D2 is produced commercially by irradiation of yeast. Vitamin D is like hormone rather than a true vitamin. The structure of vitamin D3, biologically active form of vitamin D, is similar to that of other steroid hormones, and it promotes mineral mobilization in bone and activation of osteoblasts and osteoclasts or calcium absorption from the gut. Consequently, the vitamin D deficiency leads to lose the existing bone mineral content and makes newly formed bone un- or under-mineralization excessively, osteomalacia can occur.[3] During pregnancy and lactation, maternal body requires significant amounts of calcium to pass on fetus and suckling neonate, so daily requirement of vitamin D is increased. In this manner, vitamin D deficiency would seem to be especially critical during lactation and can do as risk factor of osteomalacia.[4] A case of osteomalacia caused by prolonged lactation with femoral neck fracture was reported in 2013,[5] but there is no such kind of case in Korea. We recently diagnosed a young female as possible osteomalacia caused by prolonged lactation with vitamin D deficiency, so would like to report the case and review.
CASE
1. Demography: Thirty-five-year-old female
Chief complaint
Generalized bone pain, increased fatigability, progressive gait difficulty.
Current history
The patient is a primiparous female and had one previous vaginal delivery at term 7 years ago. She had been breastfeeding her child until who became 5-year-old. After some point, her menstrual period suddenly stopped and cannot breastfeed anymore. She was diagnosed as severe osteoporosis (Z-score from her lumbar spine was - 5.8) with L1 spinal compression fracture in regular health check-up 2 years ago. Since then she had suffered from lower back pain requiring oral analgesia, but had not received any drug treatment for osteoporosis.
Past medical history
The patient was diagnosed as active stage of pulmonary tuberculosis 9 months ago, and was treated with the standard 4 anti-tuberculosis medication (isoniazid, rifampin, ethambutol, pyrazinamide) for 6 month. She is in complete remission state now.
Family history
No one in the patient's family had past history of any disease.
Social history
The patient was a non-smoker, non-drinker, and a housewife who usually stay at home since in healthy condition. Lately she became more inactive and to be a bed-ridden state due to progressive gait difficulty. She had been lost her appetite severely and prefers to eat vegetable than meat due to abdominal discomfort since taking anti-tuberculosis medications.
Physical examination
She was 1.62 m tall and weighed 31 kg with a body mass index (BMI) of 11.81 kg/m2. Before marriage, in twenties, her body weight was approximately 45 kg and gradually decreased after child birth 7 years ago. She claimed severe pains in low back and pelvic girdle area when trying to walk. She showed antalgic lean posture and walked with a waddling gait. Besides midline tenderness over the body of her lumbar spine, marked atrophies of calf muscles with both pretibial pitting edema were noted.
Laboratory examination findings
Table 1 shows peripheral blood examination. Leukocyte, hemoglobin and platelet counts of the patient were within normal range. Also the levels of serum aspartate transaminase (AST), alanine aminotransferase (ALT), blood urea nitrogen (BUN), creatinine, sodium, potassium and chloride were all within normal range. Hypocalcemia, hypophosphatemia, high alkaline phosphatase, very low vitamin D3, high PTH, and high C-telopeptide were seen.
Radiologic findings
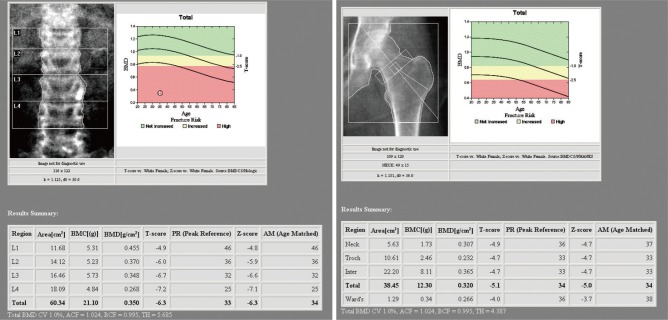
The anteroposterior and lateral radiograph of lumbar spine showed an anterior wedging with compression fracture of the L1 body and mild diffuse osteoarthritic change in facet joints (Fig. 1). Except for diffuse osteopenia, no visible pseudofracture (often called Looser zones) was showed in radiographs of pelvis (Fig. 1). The bone mineral density (BMD) value that Z-score from her lumbar spine (-6.5) and femur (-5.0) were measured by dual energy X-ray absorptiometry (DXA) (Fig. 2).
Treatment and clinical progress
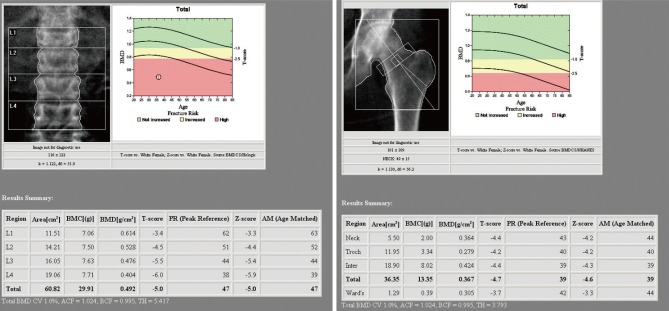
The patient was diagnosed as low BMD with vitamin D deficiency caused by prolonged lactation and malnutrition. Then we considered the possibility of osteomalacia. She was treated with oral calcium daily and 50,000 IU of Vitamin D3 once a week for 8 weeks. The condition of the patient was improved gradually, and after 8 weeks, bone and articular pain were much relieved and now she can walk without difficulty. After 12 months later, serum calcium, phosphorus, alkaline phosphatase, and vitamin D3 levels were all normalized (Table 1). Also followed up BMD showed bone mass in lumbar spine (Z-score: -5.3) and femur (-4.6) (Fig. 3).
DISCUSSION
Osteomalacia is an end-stage bone disease of severe vitamin D or phosphate depletion with characteristic biochemical, radiological and bone histologic features.[1] Definite diagnosis of osteomalacia is confirmed by bone biopsy. But measurement of plasma calcium, phosphate and alkaline phosphatase is commonly carried out as a diagnostic test for osteomalacia.[6] In addition to biochemical tests, patient's history and clinician's suspicion for disease facilitate the diagnosis of osteomalacia. For example, hypocalcemia, hypophosphatemia and high alkaline phosphatase in the presence of low serum vitamin D3 and high PTH level make the diagnosis of osteomalacia highly likely.[1] However, despite advances in biochemical tests, none of them are specific. Even normal biochemical results can be seen in histologically confirmed osteomalacia.[6]
Vitamin D deficiency is the most common cause of osteomalacia worldwide and can be induced by extrinsic or intrinsic factor either. Vitamin D3, one of the main forms of vitamin D, can obtain from sunlight exposure and also take from foods like fatty fish, milk, egg yolk, and liver oil etc. After vitamin D3 being stored in fat tissue, it acts like "back-up system" by releasing from fat in case of absence of sunlight exposure.[2] Decreased sunlight exposure by using of veil or sunscreens, inadequate dietary vitamin D intake and malabsorption caused by various gastrointestinal disorders can cause the lack of vitamin D.[1] Prolonged severe vitamin D depletion progress to hypovitaminosis D osteopathy by progressive bone turnover and defective bone mineralization. Without vitamin D, only 10% to 15% of dietary calcium and about 60% of phosphorus are absorbed.[7] Finally, irreversible cortical bone loss happens with excessive osteoid accumulation and osteomalacia occurs.
In our case, the patient had some risk factors that can lead to possible osteomalacia. First, she had a history of prolonged lactation for 5 years. During lactation, bone reabsorption is in progress due to PTH-related protein (PTHrP) production. The baby suckling activates maternal prolactin release. Both suckling and prolactin inhibit the hypothalamic gonadotropin-releasing hormone (GnRH) center. It leads to low levels of the sex steroids, estradiol, and progesterone by suppressing the gonadotropins, luteinizing hormone (LH) and follicle stimulating hormone (FSH). Only during lactation, the calcium receptor that expressed in mammary tissue controls PTHrP production and releasing. PTHrP enters the bloodstream and combines with low estradiol concentrations to up-regulate bone reabsorption. Besides maternal calcium loss is 4 times higher than in pregnancy. In fact, calcium demands are increased to accommodate the requirement of the baby, so approximately 150 to 300 mg/day of calcium loss can occur through breast milk.[8] However, there are physiological mechanisms to keep the balance of calcium and vitamin D levels in maternal body. The glomerular filtration rate (GFR) decreases during lactation, the renal excretion of calcium is reduced and calcium reabsorption from intestinal tract is happened.[8] In general, the consequence of the reabsorption of calcium from bone during lactation has been estimated that losses are 5% to 14% of BMD and during weaning, bone mass rapidly returns to normal by 0.5% to 2% a month.[9] However, this compensation mechanisms are not sufficient to fulfill the calcium loss from breast milk especially in women who already has vulnerable factors for vitamin D insufficiency. Therefore, like our case, calcium mobilization from maternal bone accelerates and the greater losses occurred in the spine and femoral neck.
Second, we can find the other risk factors for vitamin D deficiency in her life-style. She usually stays at home and became more inactive due to compression fracture of lumbar spine occurred 2 years ago. It could lead to the lack of sunlight exposure for a long time. Also inadequate intake of vitamin D-containing foods might be the other risk factor. Since in her teenage years, she has had a predilection for starchy vegetables and laid off eating protein and fat to manage her figure. During pregnancy and lactation, imbalanced food intake had been continued. Furthermore, she had been lost her appetite severely due to abdominal discomfort after taking anti-tuberculosis medications, so under-nutrition had been aggravated. Also we take notice of pulmonary tuberculosis in this patient. Several studies found out the relationship between vitamin D deficiency and pulmonary tuberculosis since 1800s.[10] Mycobacterium tuberculosis is an intracellular organism that targeting macrophages as primary host cells. Vitamin D3 is known to regulate innate immunity by interacting with macrophages,[11] therefore vitamin D deficiency can be a major risk factor for pulmonary tuberculosis by suppressing protective mechanism.
In summary, pre-existing imbalanced foods intake and prolonged lactation might lead to vitamin D insufficiency and osteoporosis in her first admission. Since then, severe back pain induced immobilization could cause the lack of sunlight exposure. Combined with inadequate supplement vitamin D, vitamin D deficiency was happened. Depletion of vitamin D increased susceptibility to Mycobacterium tuberculosis infection. On the other hand, taking the anti-tuberculosis medication caused GI trouble in this patient, so dietary intake of vitamin D was more decreased. Meanwhile, cortical bone loss continued and finally osteomalacia occurred. Our patient was in advanced stage of vitamin D deficiency, 50,000 IU of vitamin D3 once a week for 8 weeks are recommended.[1] In the early stages of treatment, fast bone re-mineralization can cause a further transient reduction of plasma levels of calcium, making it necessary to take calcium per oral (1-3 g/d).[2] Also we encouraged her to have a balanced diet and do outdoor activities regularly. To avoid vitamin D intoxication, we checked the levels of calcium, phosphorus and vitamin D closely. In conclusion, osteomalacia should be considered if the patient has unexplained bone pain or muscle weakness with suspicious history that can cause the lack of vitamin D. The disease is rare but can be curable by the early detection, accurate diagnosis with proper management.
Notes
No potential conflict of interest relevant to this article was reported.