|
 |
| jbm > Volume 29(1); 2022 > Article |
|
Abstract
Bony deformities and fragility fractures in end-stage renal disease (ESRD) patients on long-term hemodialysis can be caused by either osteoporosis or chronic kidney disease-mineral and bone disorder (CKD-MBD). Correct identification of the underlying mechanism is critical since the treatment methods differ, and one treatment approach could negatively affect the other. Cervical kyphosis, severe enough to require immediate surgical treatment, can be caused by uncontrolled CKD-MBD, albeit in limited cases. This report presents the case of a 61-year-old female with an 11-year history of hemodialysis treatment and severe cervical kyphosis with myelopathy, which required 2-stage spinal surgeries. Our report calls for a careful diagnostic approach in ESRD patients with skeletal disorders, the points to consider before calcium replacement, and early detection of fragility fractures in them. Moreover, early mobilization and weight-bearing after the surgical procedure may lead to better neurological and functional improvements.
As medical therapy improves, patients with end-stage renal disease (ESRD) enjoy longer lifespans but accompanying skeletal complications have emerged with long-term hemodialysis. Chronic kidney disease-mineral and bone disorder (CKD-MBD) is a metabolic bone diseases seen in ESRD, characterized by bone mineralization deficiency due to electrolyte and endocrine abnormalities.[1] CKD-MBD can cause not only degenerative pathology but also destructive spondyloarthropathy (DSA), most commonly at the lower cervical and lumbar spines.[2] Since CKD-MBD only accounts for less than 6% of cervical kyphosis, [3] it is often overlooked as one of the causes. In this case report, we present a female cervical kyphosis patient with a history of 11-year hemodialysis and highlight the importance of diagnosing CKD-MBD. To our knowledge, CKD-MBD was reported as the main reason for cervical kyphosis which was severe enough to cause myelopathy and to require cervical spinal fusion in only a few cases. Thus, we outline the diagnostic approach, the danger of calcium replacement, and rehabilitative considerations specifically designed for CKD-MBD patients.
A 61-year-old female first visited the neurosurgery outpatient clinic due to posterior neck pain and muscle weakness in both upper and lower extremities that had been continuous for 4 years. She was diagnosed with ESRD 11 years ago and had been undergoing hemodialysis 3 times per week. She demonstrated stage 5D CKD, evidenced by high levels of creatinine (Cr; 4.76 mg/dL; normal range, 0.55-1.02 mg/dL) and low levels vitamin D3 (25.30 ng/mL; normal range, >30 ng/mL) and glomerular filtration rate (9 mL/min/1.73m2). Until an admission for surgical correction of cervical kyphosis, she had never tested for CKD-MBD nor osteoporosis.
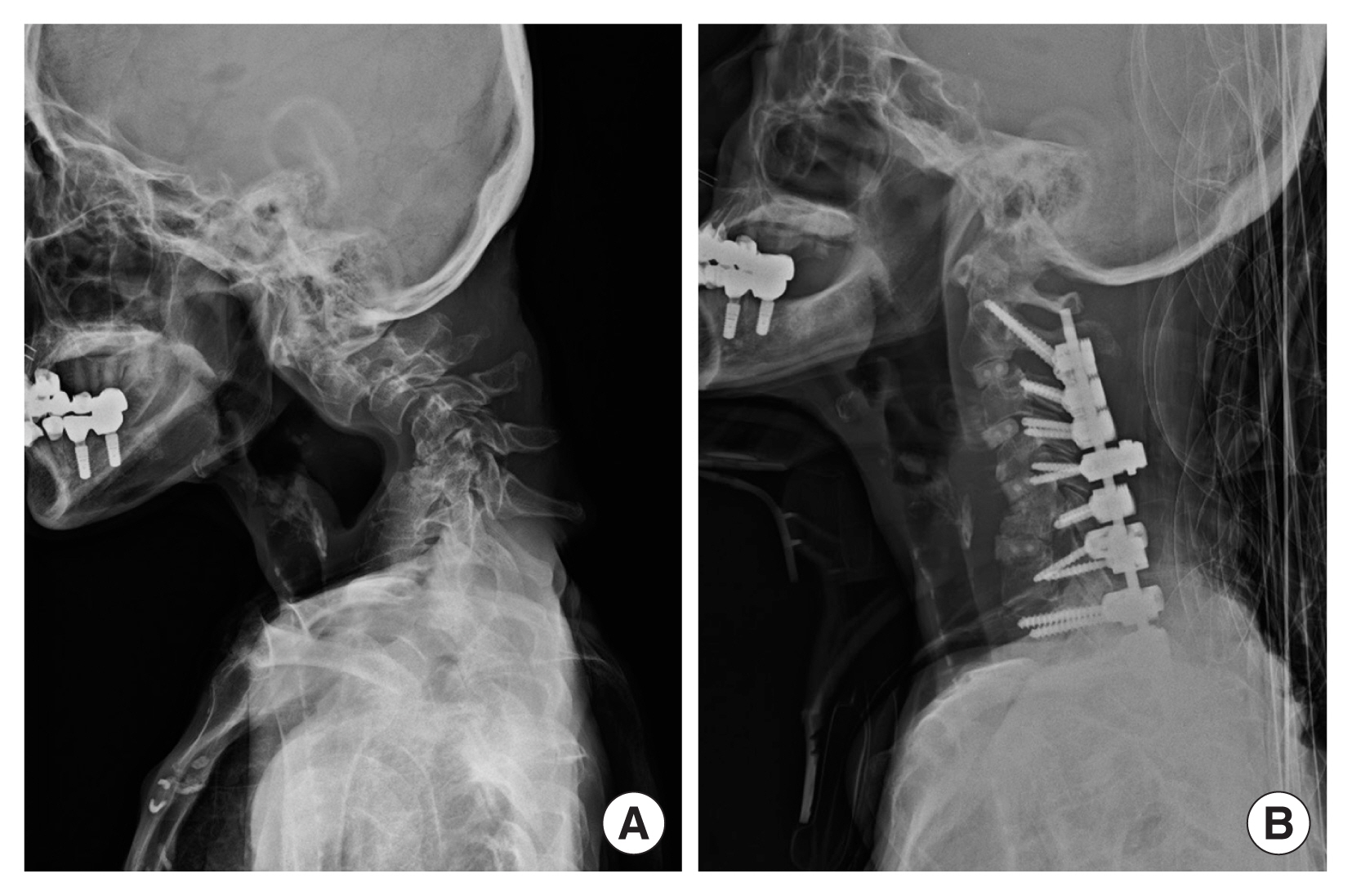
Preoperative cervical spine plain-film radiograph showed +67.0° kyphotic cervical Cobb angle as measured from the C2 vertebra to the C7 vertebra (Fig. 1A). Cervical Cobb angle was used throughout the follow-up evaluations since it is the simplest and most commonly used method to determine cervical lordosis.[4] She showed severe bilateral muscle atrophy and weakness of both upper and lower extremities, as evidenced by the Medical Research Council (MRC) muscle grade of 1 to 2.
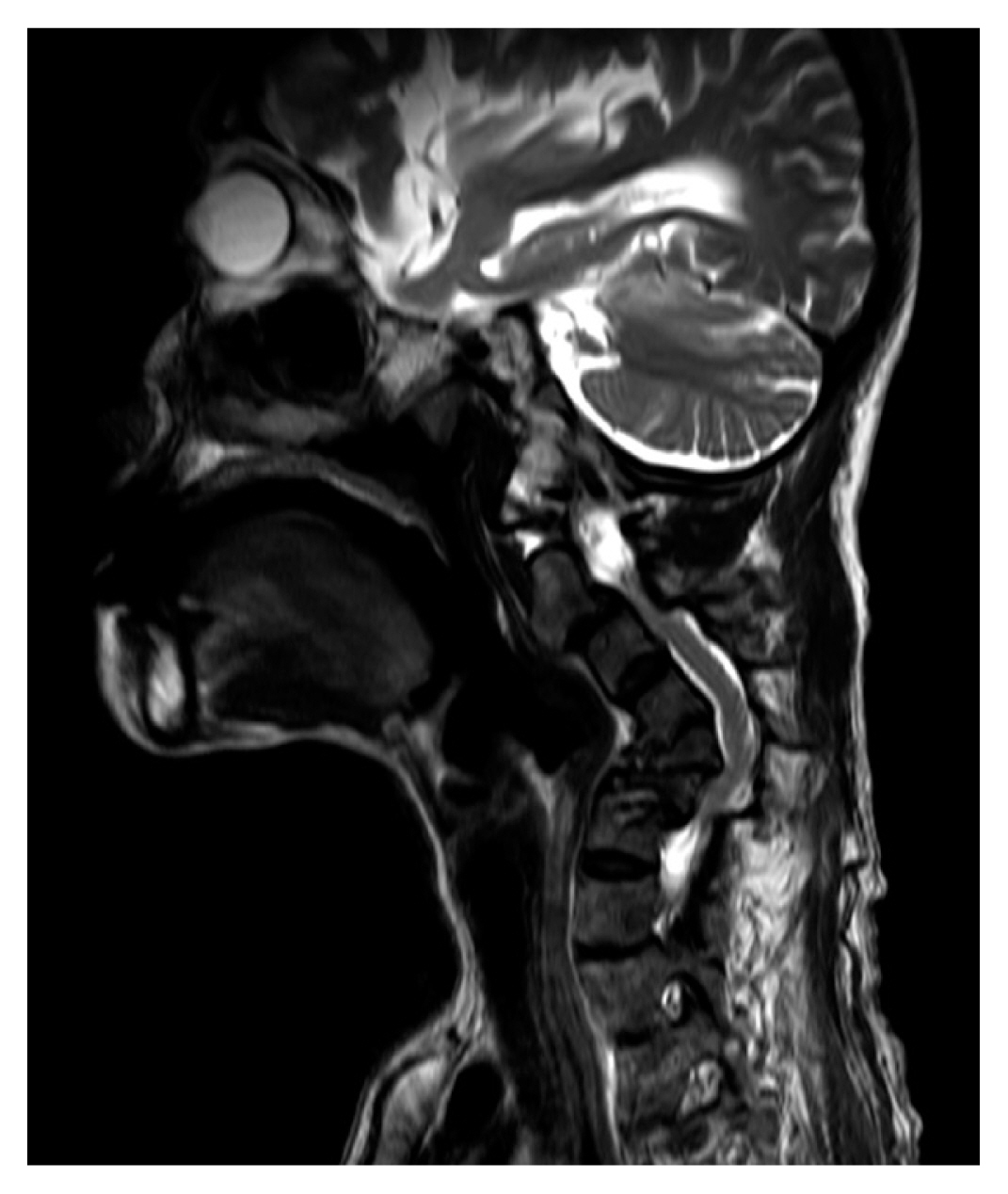
Cervical spine magnetic resonance imaging (MRI) showed severe kyphosis of the cervical spine and spinal cord compression at C3, C3-4, and C5-6 levels (Fig. 2). The C5 and C6 vertebral bodies were almost collapsed. Muscle weakness of both upper and lower extremities deteriorated to MRC muscle grade of 1. Preoperative bone mineral density (BMD) test showed osteoporosis with T-score of −3.6 in left femur head and −2.5 in lumbar spine.
To correct cervical kyphosis and myelopathy, she underwent lateral mass screw fixation at C4, 5, 6, 7 and pedicle screw fixation at C2, 7, T1, 2. Furthermore, anterior cervical discectomy and fusion was performed at C2-3-4-5 and C6-7 levels. Cervical spine plain-film radiograph after the surgery showed −9.9° lordotic cervical Cobb angle (Fig. 1B). After the operations, she has prescribed a Minerva orthosis and a complex tablet of calcium carbonate and cholecalciferol (Dicamax®; Dalim BioTech, Seoul, Korea) and teriparatide for osteoporosis management.
She was transferred to the rehabilitation department for intensive rehabilitative therapy. Since she was ESRD patient on hemodialysis, CKD-MBD was a possible reason for DSA. Endocrinologic labs showed low levels of phosphate (2.5 mg/dL; normal range, 2.9-4.6 mg/dL) and parathyroid hormone (PTH; 17.3 pg/mL; normal range, 15-65 pg/mL) and high level of N-terminal telopeptide of collagen type I (308.7 nM bone collagen equivalent [BCE]/mM Cr; normal range, 26-124 nM BCE/mM Cr). She was clinically diagnosed with adynamic bone disease, characterized by fragility fractures not explained by other etiologies, bone pain, and vascular calcifications. Thus, she was recommended not to take a calcium supplement, which can further suppress PTH.
At 1 month after the surgery, motor weakness improved to MRC grade 3 to 4 for bilateral upper and lower extremities. She was able to maintain a dynamic sitting balance and the therapist continuously provided standing experience.
Kuntz et al. [5] first reported DSA in 10 patients on long-term hemodialysis. With improved hemodialysis in ESRD, skeletal complications due to CKD-MBD will be increasingly prevalent.[6] However, discriminating fractures caused by osteoporosis from fractures by CKD-MBD presents a challenge due to the non-applicability of traditional osteoporosis diagnostic criteria.[1] In ESRD patients, osteoporosis can be diagnosed only after excluding CKD-MBD as the cause of low BMD test results or fragility fractures.
Since the early diagnosis of CKD-MBD is the most important step in managing DSA patients on hemodialysis, medical history was extensively inquired. Posterior neck pain and accompanying muscle weakness started 4 years ago and imaging studies did not show a delay in fracture healing nor recurrent stress fractures, eliminating both early and late-onset congenital causes. None of the family members showed spinal abnormality to suggest genetic causes and she never underwent other spinal surgeries before. Electromyography study did not point to motor neuron diseases and blood lab results did not indicate inflammatory diseases. Moreover, she was not involved in traumatic events and MRI did not show tumor of the spinal cord or spondylosis.
DSA in this case was caused solely by CKD-MBD. Adynamic bone disease, a subtype of CKD-MBD, can be diagnosed with a bone biopsy, but serologic markers of low bone turnover, fractures not explained by other etiologies, suppressed phosphate and PTH levels are sufficient. Since mineral homeostasis of phosphorus and calcium are disrupted in ESRD patients on hemodialysis, accompanying imbalance of hormones, including PTH, calcifediol, and calcitriol, ensues. When low bone turnover and reduced number of osteoblasts and osteoclasts cause adynamic bone disease, bone remodeling is disrupted and results in bone fractures and can lead to cervical kyphosis.[7]
CKD-MBD was diagnosed later in this case due to a hasty interpretation of the BMD test results. After identifying T-score of −3.6 in left femur head, a complex tablet of calcium carbonate and cholecalciferol was prescribed for osteoporosis management. When a calcium and cholecalciferol supplement is given to a patient with undiagnosed adynamic bone disease, PTH is further suppressed and can worsen mineral and bone disorders. Rather, the use of teriparatide, a PTH analog, may improve bone turnover in adynamic bone disease which is caused by both PTH resistance and deficiency.[8]
Currently, not many active measures are possible to manage adynamic bone disease, but calcium and vitamin D receptor activators must be avoided. Instead, prevention of DSA in CKD-MBD is more important and we recommend yearly evaluation of osteoporosis and fragility fractures since the onset of hemodialysis. The Korean National Health Insurance recommends BMD tests to 65 years or above female and 70 years or above male. The patient in our case would have benefited from early detection of bony deformities caused by long-term hemodialysis and an effort to find a true cause. Furthermore, early mobilization and weight-bearing after surgery are suggested to prevent deterioration of skeletal disorders and disuse weakness.
The limitation of our case report is the absence of whole-body fragility fracture and calcification evaluations. The most common fracture site of CKD-MBD patients is the hip, [9] followed by vertebrae, radius, clavicle, patella, and scapula.[10] Since abdomen-pelvis computed tomography (CT) showed numerous tiny calyceal stones, arterial wall calcifications in both atrophied kidneys, and calcified stones in gallbladder, further imaging evaluations, such as a whole-body bone scan or aorta CT, would have helped to locate other complications besides the cervical spine. But she was discharged shortly after the diagnosis of CKD-MBD due to a personal matter.
In conclusion, our case report showed that severe cervical kyphosis which requires surgical correction could occur as a result of uncontrolled adynamic bone disease. Since the diagnosis of osteoporosis in ESRD is different from a typical patient group, it is essential to correctly diagnose CKD-MBD in case of low BMD test results. Moreover, early CKD-MBD and fragility fractures at common sites are crucial to prevent severe DSA. Lastly, post-surgical rehabilitation must be carried out at an early stage, with an emphasis on weight bearing and mobilization.
Fig. 1
(A) Lateral upright plain-film radiograph of the cervical spine preoperatively demonstrating severe kyphosis with collapsed C5 and C6 vertebral bodies. (B) Lateral upright plain-film radiograph of the cervical spine obtained 3 weeks postoperatively demonstrating the revision of cervical kyphosis.
REFERENCES
1. Miller PD. Chronic kidney disease and the skeleton. Bone Res 2014;2:14044.https://doi.org/10.1038/boneres.2014.44.



2. Chikawa T, Sakai T, Bhatia NN, et al. Clinical outcomes of spinal surgery in patients treated with hemodialysis. J Spinal Disord Tech 2013;26:321-4. https://doi.org/10.1097/BSD.0b013e318246af73.


3. O’Shaughnessy BA, Liu JC, Hsieh PC, et al. Surgical treatment of fixed cervical kyphosis with myelopathy. Spine (Phila Pa 1976) 2008;33:771-8. https://doi.org/10.1097/BRS.0b013e3181695082.


4. Gadia A, Shah K, Nene A. Cervical kyphosis. Asian Spine J 2019;13:163-72. https://doi.org/10.31616/asj.2018.0086.


5. Kuntz D, Naveau B, Bardin T, et al. Destructive spondylarthropathy in hemodialyzed patients. A new syndrome. Arthritis Rheum 1984;27:369-75. https://doi.org/10.1002/art.1780270402.


6. Elder BD, Petteys RJ, Sciubba DM, et al. Challenges of cervical reconstruction for destructive spondyloarthropathy in renal osteodystrophy. J Clin Neurosci 2016;30:155-7. https://doi.org/10.1016/j.jocn.2016.02.005.


7. Giamalis P, Economidou D, Dimitriadis C, et al. Treatment of adynamic bone disease in a haemodialysis patient with teriparatide. Clin Kidney J 2015;8:188-90. https://doi.org/10.1093/ckj/sfv005.



8. Sista SK, Arum SM. Management of adynamic bone disease in chronic kidney disease: A brief review. J Clin Transl Endocrinol 2016;5:32-5. https://doi.org/10.1016/j.jcte.2016.07.002.



9. Maravic M, Ostertag A, Torres PU, et al. Incidence and risk factors for hip fractures in dialysis patients. Osteoporos Int 2014;25:159-65. https://doi.org/10.1007/s00198-013-2435-1.


10. Li C, Chen XM, Li Y, et al. Factors and outcome of renal osteodystrophy-associated initial fragility fracture in end-stage renal disease patients. Kidney Dis (Basel) 2019;5:118-25. https://doi.org/10.1159/000494924.



- TOOLS
-
METRICS

-
- 0 Crossref
- 0 Scopus
- 2,821 View
- 65 Download
- ORCID iDs
-
Jong Weon Lee

https://orcid.org/0000-0003-0361-5986Suhyun Cho

https://orcid.org/0000-0003-1006-5011Kyung-Min Kim

https://orcid.org/0000-0002-9825-1291Jung Hyun Park

https://orcid.org/0000-0003-3262-7476 - Related articles




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print