|
 |
| jbm > Volume 28(3); 2021 > Article |
|
Abstract
Background
Multiple members of the transforming growth factor-β (TGF-β) superfamily have well-established roles in bone homeostasis. Anti-Müllerian hormone (AMH) is a member of TGF-β superfamily of glycoproteins that is responsible for the regression of fetal Müllerian ducts and the transcription inhibition of gonadal steroidogenic enzymes. However, the involvement of AMH in bone remodeling is unknown. Therefore, we investigated whether AMH has an effect on bone cells as other TGF-β superfamily members do.
Methods
To identify the roles of AMH in bone cells, we administered AMH during osteoblast and osteoclast differentiation, cultured the cells, and then stained the cultured cells with Alizarin red and tartrate-resistant acid phosphatase, respectively. We analyzed the expression of osteoblast- or osteoclast-related genes using real-time polymerase chain reaction and western blot.
The balance between bone-resorbing osteoclasts and bone-forming osteoblasts is essential for the maintenance of bone homeostasis.[1] The imbalance in these bone cells’ activity causes various bone diseases such as osteoporosis, rheumatoid arthritis, paget’s disease, and osteopetrosis.[2] Osteoporosis is characterized by low bone mass, micro-architectural deterioration of bone tissue and consequent increase in fracture risk is caused by excessive bone resorption relative to bone formation.[3,4] While osteopetrosis is characterized by sclerotic and thick but weak and brittle bones is caused by the failure of osteoclasts to resorb bone.[5]
Multiple hormones, growth factors, and cytokines are involved in bone remodeling through regulating the proliferation, differentiation, and activation of bone cells as well as the communication between them.[6,7] In particular, the growth factors belonging to the transforming growth factor β (TGF-β) superfamily play an important role in the balance between bone formation and resorption through directly or indirectly regulating osteogenesis and osteoclastogenesis.[6,8] There are several subfamilies of TGF-β superfamily including the TGF-β, Nodal, Activin subfamily, the bone morphogenetic protein (BMP), growth/differentiation factor (GDF) family, and the group of anti-Müllerian hormone/Müllerian inhibiting substance (AMH/MIS).[6,8]
TGF-β/BMPs have absolutely recognized roles in bone formation both in vivo and in vitro.[6,8,9] Especially, BMP-2, BMP-4, BMP-6, and BMP-9 induce osteoblastic differentiation and bone formation, in addition, BMP-2 and BMP-7 in combination further enhance osteoblastic differentiation than that induced by BMP-2 or BMP-7 alone.[10-15] TGF-β1 also drastically enhances ectopic bone formation induced by BMP-2.[15] The roles of TGF-β/BMPs in bone resorption are relatively less established compared to bone formation and still controversial. BMP-7, and BMP-9 promote receptor activator of nuclear factor-κB (NF-κB) ligand (RANKL)-induced osteoclast differentiation.[16,17] While BMP-2, TGF-β1 and TGF-β2 have a biphasic effect on osteoclast differentiation concentration- or stage of differentiation-dependently.[18-20]
AMH, also known as MIS, is a dimeric glycoprotein exclusively expressed by granulosa cells of pre-antral and small antral follicles in the ovary. AMH plays an important role in chronic anovulation by inhibiting the initial and cyclic recruitment of ovarian follicles and by promoting follicular arrest.[21,22] In addition, AMH has antiproliferative effects on ovarian and breast tumor cell lines.[21,23] Since AMH is a distinct member of the TGF-β superfamily, it is expected that AMH signaling is similar to that of the well-defined signaling pathway of other TGF-β family members.[21] However, despite most family members of TGF-β superfamily are involved in a vast majority of cellular processes and thereby exhibiting versatile functions in the body, the known function of AMH is still restricted to the reproductive organs.[6,21,24] In the present study, we investigated whether 2 types of bone cells are target cell(s) to translate the signals of AMH, and AMH regulates osteogenesis and osteoclastogenesis like other TGF-β family members.
Recombinant human RANKL was purified from bacteria. Recombinant human macrophage colony-stimulating factor (M-CSF) was a gift from Dr. Daved Fremont (Washington University, St. Louis, MO, USA). Recombinant human BMP2 protein was purchased from Cowellmedi (Busan, Korea). Ascorbic acid was purchased from Junsei Chemical (Tokyo, Japan). β-glycerophosphate was purchased from Sigma-Aldrich (St. Louis, MO, USA). Recombinant mouse AMH was purchased from Cloud-Clone Crop. (Wuhan, China).
Primary osteoblast precursor cells were isolated from newborn of ICR mouse calvaria via enzymatic digestion with 0.1% collagenase (Life Technologies, Carlsbad, CA, USA) and 0.2% dispase II (Roche Diagnostics, Mannheim, Germany). Primary osteoblast precursor cells were cultured in an osteogenic medium containing BMP2 (100 ng/mL; Cowellmedi), ascorbic acid (50 μg/mL; Junsei Chemical), and β-glycerophosphate (100 mM; Sigma-Aldrich) for 3 or 6 days. Cultured cells for 3 days were lysed using osteoblast lysis buffer (50 mM Tris-HCl [pH 7.4], 1% Triton X-100, 150 mM NaCl, and 1 mM ethylenediaminetetraacetic acid [EDTA]). Cell lysates were incubated with p-nitrophenyl phosphate substrate (Sigma-Aldrich), and alkaline phosphatase (ALP) activity was determined by measuring the absorbance at 405 nm using a spectrophotometer. Cultured cells for 6 days were fixed with 70% ethanol and stained with 40 mM Alizarin red (pH 4.2). After removing nonspecific staining with phosphate buffered saline, stained cells were visualized with a CanoScan 4400F (Canon Inc., Tokyo, Japan). Alizarin red-stained cells were incubated with 10% cetylpyridinium chloride solution for 30 min at room temperature, and the absorbance was determined at 562 nm.
Mouse bone marrow cells were isolated from tibiae and femurs of 6-week-old ICR mice by flushing the bone marrow with α-minimal essential medium (α-MEM; Hyclone Lab Inc., Logan, UT, USA). Bone marrow cells were cultured for 3 days in α-MEM (Hyclone Lab Inc.) containing 10% fetal bovine serum (FBS; Hyclone Lab Inc.) in the presence of M-CSF (30 ng/mL). Adherent cells were further cultured in the presence of M-CSF (30 ng/mL) and RANKL (150 ng/mL) for 3 days. Cultured cells were fixed with 3.7% formalin and stained for tartrate-resistant acid phosphatase (TRAP). TRAP-positive cells with more than 3 nuclei were considered TRAP-positive MNCs. Cells were observed using a Leica DMIRB microscope equipped with an N Plan 10×0.25 numerical aperture objective lens (Leica Microsystems, Wetzlar, Germany). Images were captured with a ProgRes CFscan camera (Jenoptik, Jena, Germany) using ProgRes Capture Pro software (Jenoptik).
Osteoclast precursor cells were cultured for 3 days in α-MEM (Hyclone Lab Inc.) containing 10% FBS (Hyclone Lab Inc.) in the presence of M-CSF (30 ng/mL) in a 96-well plate. The cell proliferation was measured with the MTT kit (Sigma-Aldrich) according to the manufacturer’s protocol.
Total RNA was prepared using the Qiazol reagent (Qiagen, Valencia, CA, USA), and 2 μg of the isolated RNA was reverse transcribed into cDNA using Superscript II Reverse Transcriptase (Invitrogen; Thermo Fisher Scientific, lnc., Waltham, MA, USA). Quantitative real-time PCR analysis was performed in triplicate with a Rotor-Gene Q (Qiagen) with SYBR Green (Qiagen). Expression levels were normalized against endogenous glyceraldehyde 3-phosphate dehydrogenase levels. The following primers were used: Gadph, 5′-TGA CCA CAG TCC ATG CCA TCA CTG-3′ and 5′-CAG GAG ACA ACC TGG TCC TCA GTG-3′; cfos, 5′-ATG GGC TCT CCT GTC AAC ACA-3′ and 5′-TGG CAA TCT CAG TCT GCA ACG CAG-3′; Nfatc1, 5′-CTC GAA AGA CAG CAC TGG AGC AT-3′ and 5′-CGG CTG CCT TCC GTC TCA TAG-3′; Acp5, 5′-CTG GAG TGC ACG ATG CCA GCG ACA-3′ and 5′-TCC GTG CTC GGC GAT GGA CCA GA-3′; Runx2, 5′-CCC AGC CAC CTT TAC CTA CA-3′ and 5′-CAG CGT CAA CAC CAT TC-3′; Alpl, 5′-CAA GGA TAT CGA CGT GAT CAT G-3′ and 5′-GTC AGT CAG GTT CCG ATT C-3′; Ibsp, 5′-GGA AGA GGA GAC TTC AAA CGA AG-3′ and 5′-CAT CCA CTT CTG CTT CGT TC-3′; Bglap, 5′-ATG AGG ACC CTC TCT CTG CTC AC-3′ and 5′-AGA GCA AAC TGC AGA AGC TGA GAG-3′; and Amh, 5′-GGA GTC TGC AGC ACT GAC TC-3 and 5′-TCA CTT CAG CCA GAT GTA GG-3′.
Cultured cells were lysed in extraction buffer (50 mM Tris-HCl [pH 8.0], 150 mM NaCl, 1 mM EDTA, 0.5% Nonidet P-40, 1 mM PMSF, and protease inhibitor cocktail). After centrifugation, equal amounts of proteins were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred onto polyvinylidene difluoride membranes (Millipore, Billerica, MA, USA). Membranes were incubated with the appropriate antibodies against c-Fos, NFATc1, Cathepsin K, Actin (Santa Cruz Biotechnology, Dallas, TX, USA), phospho-p38, p38, phospho-c-JUN N-terminal kinase (JNK), JNK, phospho-Erk, Erk, and IκB (Cell Signaling Technology, Danvers, MA, USA). Signals were detected with an enhanced chemiluminescence reagent (Millipore) and analyzed using an Azure c300 luminescent image analyzer (Azure Biosystems, Dublin, CA, USA).
All values are expressed as the means±standard deviation. Statistical significance was determined by using 2-tailed Student’s t-tests for 2 independent samples or analysis of variance with post-hoc Tukey’s honestly significant difference test for multiple group comparisons. A P-value of less than 0.05 was considered statistically significant.
To determine whether AMH participates in osteoblast differentiation, gene expression of Amh was analyzed during the differentiation of osteoblasts. Primary osteoblast precursor cells were induced to osteoblasts by culturing in an osteogenic medium containing BMP-2, ascorbic acid, and β-glycerophosphate. It was observed that Amh gene expression was not detected in BMP-2-induced osteoblast differentiation while there were inductions of osteoblast differentiation marker genes, including Runx2, Alpl, Ibsp, and Bglap (Fig. 1A). Next, Amh gene expression was analyzed during osteoclast differentiation. Bone marrow-derived macrophages (BMMs) were differentiated to osteoclast by culturing in an osteoclastogenic medium containing M-CSF and RANKL. Despite induction of osteoclast differentiation marker genes such as c-fos, Nfatc1, and Acp5, Amh gene expression was undetectable in RANKL-induced osteoclast differentiation (Fig. 1B).
Since Amh was not expressed during osteoblast differentiation, the effect of AMH on osteoblast differentiation and function via treatment of exogeneous AMH during the BMP2-induced osteoblast differentiation of primary osteoblast precursor cells. Upon treatment of exogeneous AMH, there was no significant difference in ALP activity and nodule formation was slightly increased (Fig. 2A, B). Furthermore, AMH did not have any effect on gene expressions of osteoblast differentiation marker genes such as Runx2, Alpl, Ibsp, and Bglap (Fig. 2C). These results indicate that AMH is not involved in BMP2-induced osteoblast differentiation of primary osteoblast precursor cells.
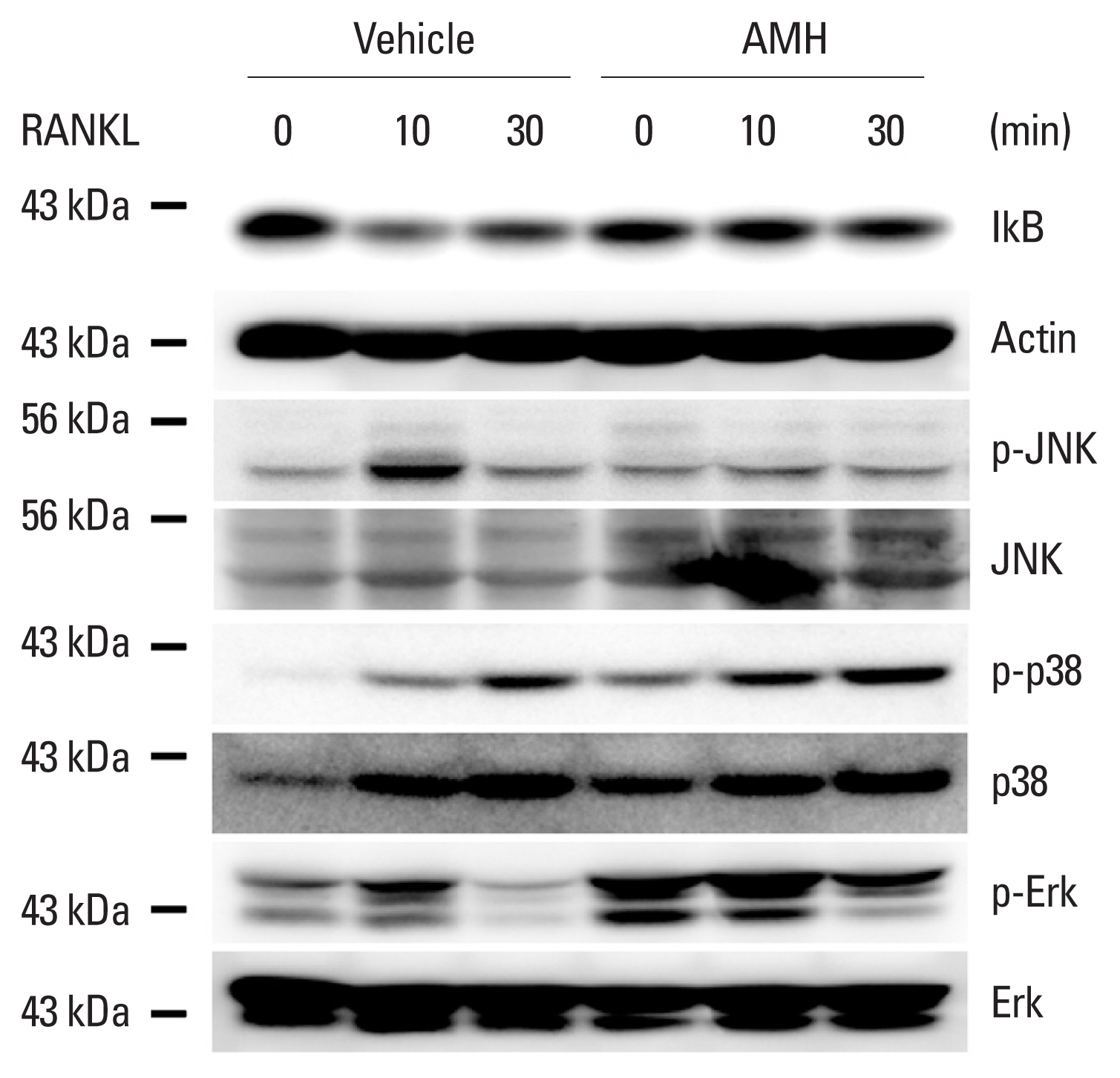
Next, the effect of AMH on RANKL-induced osteoclast differentiation of BMMs was investigated via treatment of AMH in various doses during the osteoclast differentiation. AMH treatment significantly inhibited TRAP-positive multinuclear osteoclast formation in a dose dependent manner according to TRAP stain analyses without affecting proliferation of osteoclast precursor cells (Fig. 3A). Furthermore, AMH dramatically suppressed expression levels of osteoclast differentiation markers such as c-Fos, NFATc1, and TRAP at both mRNA and protein levels (Fig. 3B, C). Since AMH inhibited osteoclast differentiation, its effect on early RANKL signaling pathway was investigated. Phosphorylation of p38 and Erk was increased by AMH pretreatment, which seems to be activated by AMH rather than affecting the signaling activated by RANKL. Among several signaling pathways activated by RANKL, JNK activation was inhibited, and above all, IκB degradation was most strongly inhibited (Fig. 4). These results suggest that the strong inhibition of IκB degradation which occur simultaneously with inhibition of JNK activation is responsible for inhibitory effect of AMH on RANKL-induced osteoclast differentiation.
AMH has been demonstrated that plays an important role in ovarian function.[22] In addition to AMH, BMPs is known for its critical role in the regulation of ovarian function, of which BMP-2, BMP-6, BMP-7, BMP-15, and GDF-9 are expressed in the human ovary, and specially, BMP-15 and GDF-9 are exclusively expressed in the oocytes.[25-29] Ogura-Nose et al. [25] showed that BMP-2, BMP-6, BMP-7, and BMP-15, but not GDF-9, induced both mRNA and protein expression of AMH in human granulosa cells. As other members of the TGF-β family, AMH first transmits signals through the formation of heteromeric complexes of specific type I and type II serine/threonine kinase receptors.[21] The activation of specific type II receptor by binding AMH is followed by phosphorylation of the type I receptor. Activated type I receptor results in phosphorylation of the downstream Smad proteins. Phosphorylated downstream Smad proteins form a complex with common Smad4 and this complex is translocated to the nucleus to regulate target gene expression.[21,30] AMH has been reported to activate BMP like a pathway through BMP type I receptors such as activin-like kinase (ALK)2 and ALK6 to activate the Smad5 and Smad1 pathways respectively.[30-32] The results imply that AMH expression is increased by BMPs and the Smad1 and Smad2 pathway are shared by the BMPs and AMH led us to test whether AMH is induced by BMP2 in osteoblasts and AMH can regulate osteoblast differentiation by activating BMP like pathway. Unexpectedly, AMH expression was not increased by BMP-2, nor AMH affect BMP-2-mediated osteoblast differentiation in primary osteoblasts indicating that osteoblasts are not target cells of AMH under the physiological condition.
AMH seems to activate the BMP-like pathway to induce Smad1-dependent signaling in osteoclast precursor cells. Josso et al. [33] reported that AMH type II receptor (AMHR-II) interacts with ALK5 and ALK6 among the type I receptors and interaction with ALK5 is ligand-independent while interaction with ALK6 is increased by AMH. They also showed that AMH activates Smad1 but not Smad2 signal transduction. Smad1 signal has been demonstrated to inhibit osteoclast differentiation. Inhibitory effect of TGF-β1 during human osteoclastogenesis is mediated by Smad1 signaling.[33] In addition, BMP-Smad1 signaling inhibits the initiation of osteoclast differentiation by suppressing the RANKL-NF-κB pathway.[20] Our results showed that AMH inhibited RANKL-induced osteoclast differentiation without affecting proliferation of osteoclast precursor cells and AMH pretreatment strongly caused a decrease in IκBα degradation medicated by RANKL. These results are consistent with that BMP2 pretreatment caused an increase in IκB and a decrease in NF-κB during M-CSF/RANKL-induced osteoclast commitment and indicating that AMH negatively regulates osteoclast differentiation by suppressing the RANKL-NF-κB pathway.
In this study, we have provided clues on the role of AMH during bone remodeling. AMH was found to inhibit osteoclast differentiation without affecting osteoblast differentiation. Further study on the role and action mechanism of AMH in osteoclasts and osteoblasts will help to understand the complex mechanisms that how TGF-β superfamily influence bone homeostasis and bone disease.
DECLARATIONS
Funding
This work was supported by National Research Foundation of Korea (NRF) grants funded by the Korean government (MSIT) (2019R1A5A2027521).
Fig. 1
Expression of Anti-Müllerian hormone (AMH) in bone cells. (A) Osteoblasts were cultured with osteogenic medium containing bone morphogenetic proteins 2, ascorbic acid, and β-glycerophosphate for the indicated times. (B) Bone marrow-derived macrophages were cultured with macrophage colony-stimulating factor (M-CSF) alone or M-CSF and receptor activator of nuclear factor-κB ligand (RANKL) for the indicated times. (A, B) Total RNA was extracted at each indicated time point. Quantitative real-time polymerase chain reaction analysis was performed to measure the mRNA expression levels of the target genes. #P<0.05. *P<0.01. **P<0.001 vs. control.

Fig. 2
Anti-Müllerian hormone (AMH) does not affect osteoblast differentiation. (A-C) Osteoblasts were cultured with osteogenic medium (OGM) in the absence or presence of AMH (100 ng/mL). (A) Cells were cultured for 3 days and subjected to alkaline phosphatase (ALP) activity assay. (B) Cultured cells for 6 days were fixed and stained with Alizarin red (left panel). Staining intensities were quantified via densitometry at 562 nm (right panel). (C) Total RNA was extracted at each indicated time point. Quantitative real-time polymerase chain reaction analysis was performed to measure the mRNA expression levels of the target genes. *P<0.01. **P<0.001 vs. control.
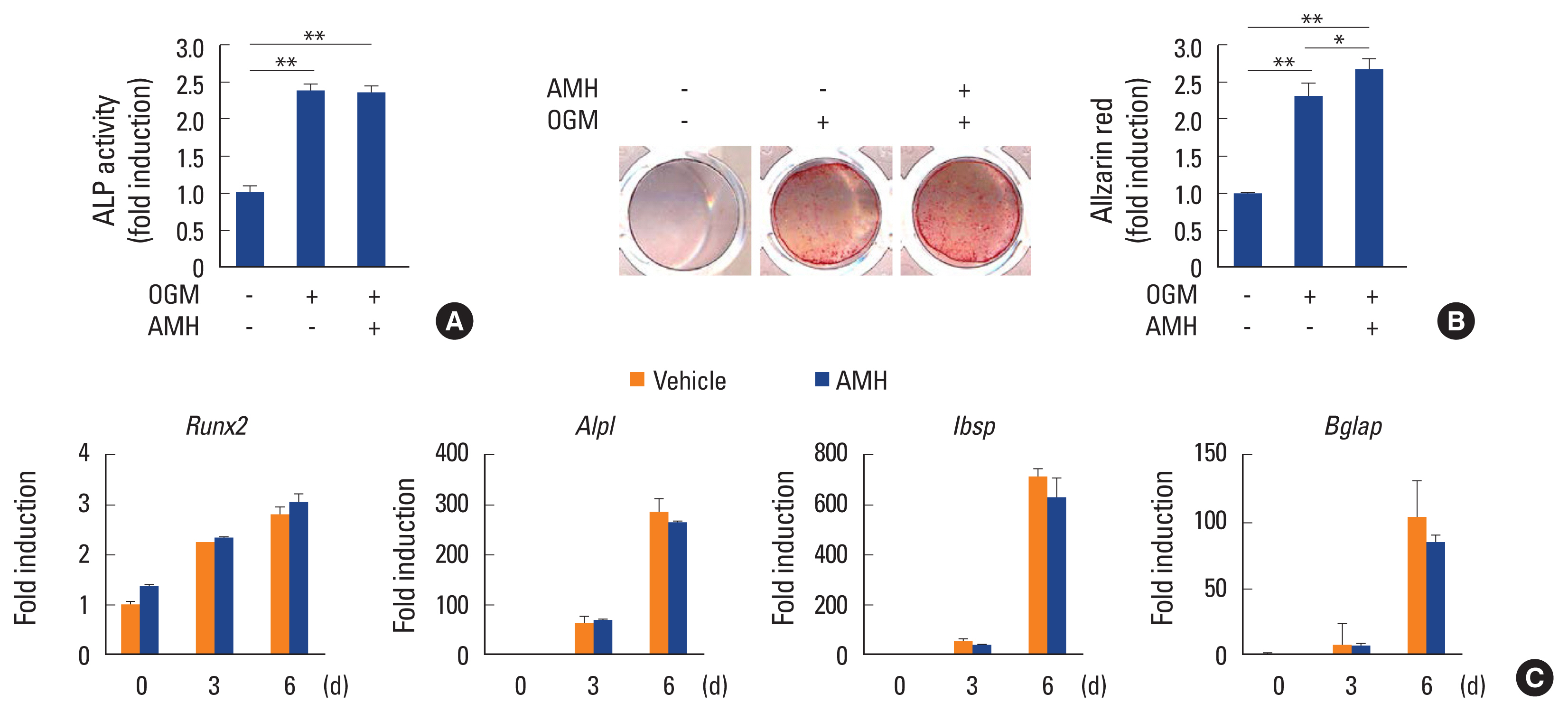
Fig. 3
Anti-Müllerian hormone (AMH) inhibits osteoclast differentiation. (A-C) Bone marrow-derived macrophages were cultured with macrophage colony-stimulating factor (M-CSF) alone or M-CSF and receptor activator of nuclear factor-κB ligand (RANKL) in the absence or presence of AMH as indicated. (A) Cultured cells were fixed and stained for TRAP (upper panel). Numbers of TRAP (+) multinucleated cells were counted (lower left panel). The cell proliferation was measured by MTT assay (lower right panel). (B) Total RNA was extracted at each indicated time point. Quantitative real-time polymerase chain reaction analysis was performed to measure the mRNA expression levels of the target genes. (C) Whole cell lysates were analyzed via western blotting using specific antibodies as indicated. #P<0.05. *P<0.01. **P<0.001 vs. control.
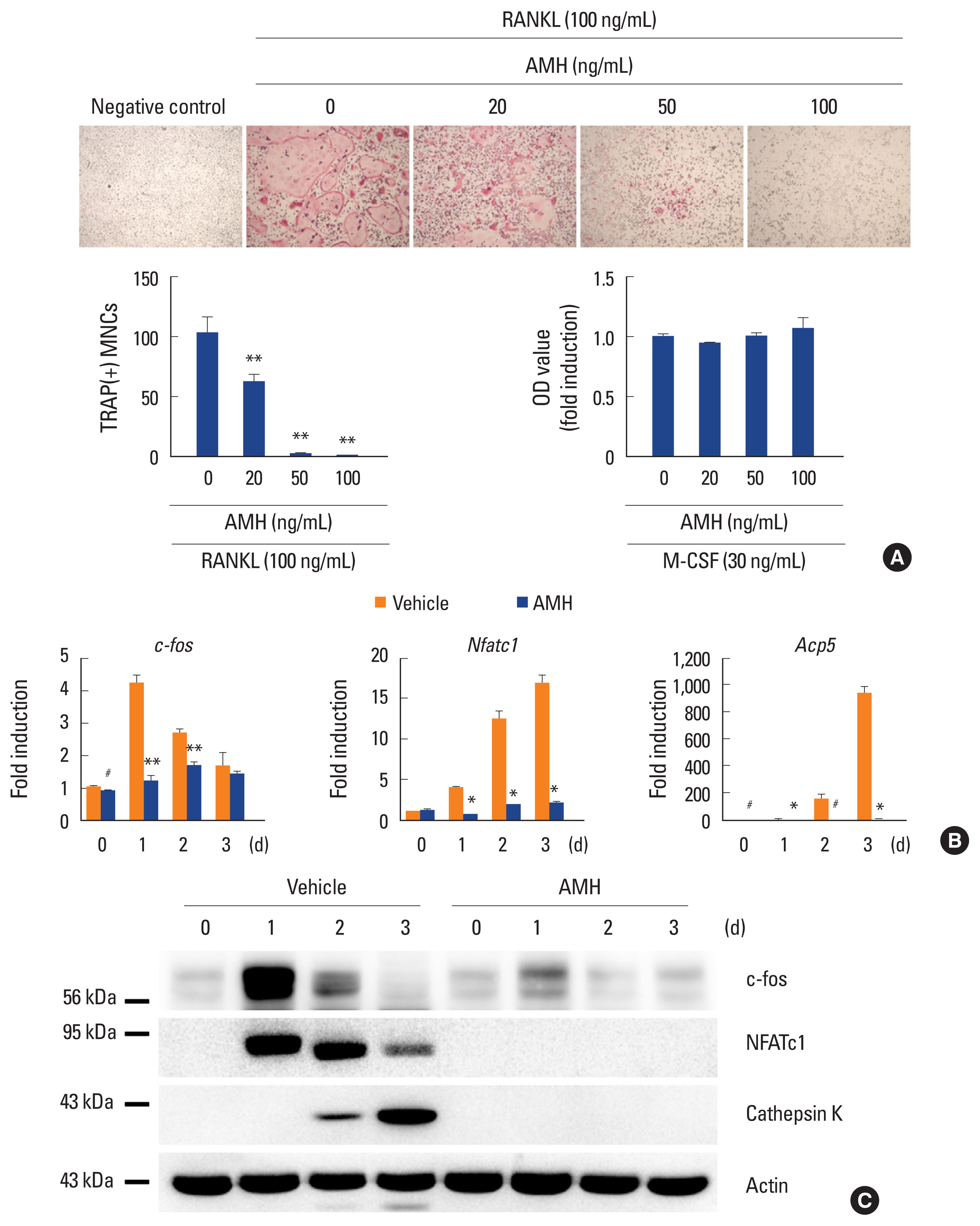
Fig. 4
Anti-Müllerian hormone (AMH) inhibits receptor activator of nuclear factor-κB ligand (RANKL)-induced IkB degradation. Bone marrow-derived macrophages were serum-starved, pretreated with vehicle or AMH for 1 hr and stimulated with RANKL (500 ng/mL) for the indicated times. Whole cell lysates were analyzed via western blotting using specific antibodies as indicated.
REFERENCES
1. Sims NA, Martin TJ. Coupling the activities of bone formation and resorption: a multitude of signals within the basic multicellular unit. Bonekey Rep 2014;3:481.
https://doi.org/10.1038/bonekey.2013.215
.



2. Sharma S, Mahajan A, Mittal A, et al. Epigenetic and transcriptional regulation of osteoclastogenesis in the pathogenesis of skeletal diseases: A systematic review. Bone 2020;138:115507.
https://doi.org/10.1016/j.bone.2020.115507
.


3. Rachner TD, Khosla S, Hofbauer LC. Osteoporosis: now and the future. Lancet 2011;377:1276-87.
https://doi.org/10.1016/s0140-6736(10)62349-5
.



4. Cotts KG, Cifu AS. Treatment of osteoporosis. JAMA 2018;319:1040-1.
https://doi.org/10.1001/jama.2017.21995
.


5. Stark Z, Savarirayan R. Osteopetrosis. Orphanet J Rare Dis 2009;4:5.
https://doi.org/10.1186/1750-1172-4-5
.



6. Jann J, Gascon S, Roux S, et al. Influence of the TGF-β superfamily on osteoclasts/osteoblasts balance in physiological and pathological bone conditions. Int J Mol Sci 2020;
https://doi.org/10.3390/ijms21207597
.

7. Walters G, Pountos I, Giannoudis PV. The cytokines and micro-environment of fracture haematoma: Current evidence. J Tissue Eng Regen Med 2018;12:e1662-77.
https://doi.org/10.1002/term.2593
.


8. Chen G, Deng C, Li YP. TGF-β and BMP signaling in osteoblast differentiation and bone formation. Int J Biol Sci 2012;8:272-88.
https://doi.org/10.7150/ijbs.2929
.



9. Linkhart TA, Mohan S, Baylink DJ. Growth factors for bone growth and repair: IGF, TGF beta and BMP. Bone 1996;19:1s-12s.
https://doi.org/10.1016/s8756-3282(96)00138-x
.

10. Cheng H, Jiang W, Phillips FM, et al. Osteogenic activity of the fourteen types of human bone morphogenetic proteins (BMPs). J Bone Joint Surg Am 2003;85:1544-52.
https://doi.org/10.2106/00004623-200308000-00017
.


11. Kang Q, Sun MH, Cheng H, et al. Characterization of the distinct orthotopic bone-forming activity of 14 BMPs using recombinant adenovirus-mediated gene delivery. Gene Ther 2004;11:1312-20.
https://doi.org/10.1038/sj.gt.3302298
.


12. Cui J, Zhang W, Huang E, et al. BMP9-induced osteoblastic differentiation requires functional Notch signaling in mesenchymal stem cells. Lab Invest 2019;99:58-71.
https://doi.org/10.1038/s41374-018-0087-7
.


13. Zhang Y, Yang W, Devit A, et al. Efficiency of coculture with angiogenic cells or physiological BMP-2 administration on improving osteogenic differentiation and bone formation of MSCs. J Biomed Mater Res A 2019;107:643-53.
https://doi.org/10.1002/jbm.a.36581
.


14. Sun P, Wang J, Zheng Y, et al. BMP2/7 heterodimer is a stronger inducer of bone regeneration in peri-implant bone defects model than BMP2 or BMP7 homodimer. Dent Mater J 2012;31:239-48.
https://doi.org/10.4012/dmj.2011-191
.


15. Tachi K, Takami M, Sato H, et al. Enhancement of bone morphogenetic protein-2-induced ectopic bone formation by transforming growth factor-β1. Tissue Eng Part A 2011;17:597-606.
https://doi.org/10.1089/ten.TEA.2010.0094
.


16. Li H, Zhao D, Wang S, et al. Bone morphogenetic protein-9 promotes the differentiation of mouse spleen macrophages into osteoclasts via the ALK1 receptor and ERK 1/2 pathways in vitro. Mol Med Rep 2016;14:4545-50.
https://doi.org/10.3892/mmr.2016.5803
.



17. Zheng Y, Wang L, Zhang X, et al. BMP2/7 heterodimer can modulate all cellular events of the in vitro RANKL-mediated osteoclastogenesis, respectively, in different dose patterns. Tissue Eng Part A 2012;18:621-30.
https://doi.org/10.1089/ten.TEA.2011.0366
.


18. Karst M, Gorny G, Galvin RJ, et al. Roles of stromal cell RANKL, OPG, and M-CSF expression in biphasic TGF-beta regulation of osteoclast differentiation. J Cell Physiol 2004;200:99-106.
https://doi.org/10.1002/jcp.20036
.



19. Lee B, Oh Y, Jo S, et al. A dual role of TGF-β in human osteoclast differentiation mediated by Smad1 versus Smad3 signaling. Immunol Lett 2019;206:33-40.
https://doi.org/10.1016/j.imlet.2018.12.003
.


20. Li A, Cong Q, Xia X, et al. Pharmacologic calcitriol inhibits osteoclast lineage commitment via the BMP-Smad1 and IκB-NF-κB pathways. J Bone Miner Res 2017;32:1406-20.
https://doi.org/10.1002/jbmr.3146
.


21. Visser JA. AMH signaling: from receptor to target gene. Mol Cell Endocrinol 2003;211:65-73.
https://doi.org/10.1016/j.mce.2003.09.012
.


22. Durlinger AL, Visser JA, Themmen AP. Regulation of ovarian function: the role of anti-Müllerian hormone. Reproduction 2002;124:601-9.
https://doi.org/10.1530/rep.0.1240601
.


23. Teixeira J, Maheswaran S, Donahoe PK. Müllerian inhibiting substance: an instructive developmental hormone with diagnostic and possible therapeutic applications. Endocr Rev 2001;22:657-74.
https://doi.org/10.1210/edrv.22.5.0445
.


24. Lee MM, Donahoe PK. Mullerian inhibiting substance: a gonadal hormone with multiple functions. Endocr Rev 1993;14:152-64.
https://doi.org/10.1210/edrv-14-2-152
.


25. Ogura-Nose S, Yoshino O, Osuga Y, et al. Anti-Mullerian hormone (AMH) is induced by bone morphogenetic protein (BMP) cytokines in human granulosa cells. Eur J Obstet Gynecol Reprod Biol 2012;164:44-7.
https://doi.org/10.1016/j.ejogrb.2012.05.017
.


26. Shi J, Yoshino O, Osuga Y, et al. Bone morphogenetic protein-6 stimulates gene expression of follicle-stimulating hormone receptor, inhibin/activin beta subunits, and anti-Müllerian hormone in human granulosa cells. Fertil Steril 2009;92:1794-8.
https://doi.org/10.1016/j.fertnstert.2009.05.004
.


27. Ross A, Munger S, Capel B. Bmp7 regulates germ cell proliferation in mouse fetal gonads. Sex Dev 2007;1:127-37.
https://doi.org/10.1159/000100034
.


28. Lawson KA, Dunn NR, Roelen BA, et al. Bmp4 is required for the generation of primordial germ cells in the mouse embryo. Genes Dev 1999;13:424-36.
https://doi.org/10.1101/gad.13.4.424
.



29. Shimasaki S, Moore RK, Otsuka F, et al. The bone morphogenetic protein system in mammalian reproduction. Endocr Rev 2004;25:72-101.
https://doi.org/10.1210/er.2003-0007
.


30. Prapa E, Vasilaki A, Dafopoulos K, et al. Effect of Anti-Müllerian hormone (AMH) and bone morphogenetic protein 15 (BMP-15) on steroidogenesis in primary-cultured human luteinizing granulosa cells through Smad5 signalling. J Assist Reprod Genet 2015;32:1079-88.
https://doi.org/10.1007/s10815-015-0494-2
.



31. Visser JA, Olaso R, Verhoef-Post M, et al. The serine/threonine transmembrane receptor ALK2 mediates Müllerian inhibiting substance signaling. Mol Endocrinol 2001;15:936-45.
https://doi.org/10.1210/mend.15.6.0645
.


32. Gouédard L, Chen YG, Thevenet L, et al. Engagement of bone morphogenetic protein type IB receptor and Smad1 signaling by anti-Müllerian hormone and its type II receptor. J Biol Chem 2000;275:27973-8.
https://doi.org/10.1074/jbc.M002704200
.


33. Josso N, di Clemente N, Gouédard L. Anti-Müllerian hormone and its receptors. Mol Cell Endocrinol 2001;179:25-32.
https://doi.org/10.1016/s0303-7207(01)00467-1
.


- TOOLS
-
METRICS

- ORCID iDs
-
Jung Ha Kim

https://orcid.org/0000-0003-2543-043XKi-Sun Kwon

https://orcid.org/0000-0002-6401-2735Nacksung Kim

https://orcid.org/0000-0002-8132-3981 - Related articles




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print